Back
Endocrinology: All areas
Category: Abstract Submission
Endocrinology I
521 - Does Introduction of Oral Dextrose Gel Pathway Decrease the Length of Stay in Neonates With Asymptomatic Hypoglycemia?
Friday, April 22, 2022
6:15 PM – 8:45 PM US MT
Poster Number: 521
Publication Number: 521.107
Publication Number: 521.107
Omar Beckett, Maimonides Infants and Children's Hospital of Brooklyn, Staten Island, NY, United States; Alok Bhutada, Maimonides Children's Hospital of Brooklyn, Brooklyn, NY, United States; Antonios Likourezos, Maimonides Medical Center, Brooklyn, NY, United States; Yelena Kogelman, Maimonides Infants and Children's Hospital of Brooklyn, Brooklyn, NY, United States

Omar Beckett, DO
Pediatric Endocrinology Fellow
Maimonides Infants and Children's Hospital of Brooklyn
Staten Island, New York, United States
Presenting Author(s)
Background: Neonatal hypoglycemia has been associated with various forms of neurological impairment including developmental delay, seizures, visual processing problems, and cognitive difficulties[1]. Consensus guidelines have been created to expedite the identification and resolution of hypoglycemic episodes. Interventions, such oral dextrose gel, have been shown to quickly and effectively reverse episodes of severe hypoglycemia [2]. However, more research needs to be conducted to quantify potential effects of oral glucose gel clinical pathways on prevention of hypoglycemia in the neonatal period.
Objective: We evaluated the effects of introducing a clinical pathway for improving hypoglycemia with oral dextrose gel. We hypothesized a decrease in the length of stay of neonates with hypoglycemia after the introduction of the clinical pathway.
Design/Methods: We conducted a retrospective chart review evaluating neonates born ≥ 35 weeks gestational age, who exhibited asymptomatic hypoglycemia within 48 hours of life (HOL) while receiving exclusive oral feeds. The primary outcome was overall length of hospital stay. We extracted data from charts of children born prior and after the implementation of this oral dextrose gel pathway in both the well-baby nursery and Neonatal Intensive Care Unit (NICU) of a single medical center over a one-year period.
Results: 385 total neonates who fit the aforementioned criteria were included, with 175 neonates representing the historical control group and 210 neonates in the intervention group receiving oral dextrose gel. Analysis demonstrated no significant difference in the overall length of hospital stay, as well as the total episodes of hypoglycemia and number of days the neonate received intravenous fluid therapy with dextrose.Conclusion(s): Our study demonstrates that the introduction of oral dextrose gel pathway did not have a significant effect on the overall length of hospital stay, as well as the total episodes of hypoglycemia and number of days the neonate received intravenous fluid therapy with dextrose at our institution. Further research is needed to evaluate whether response to oral dextrose gel can be used as a predictor of severe hypoglycemia in the neonatal period.
Table 1. Characteristics of Study Subjects.jpg) All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
Table 2. Results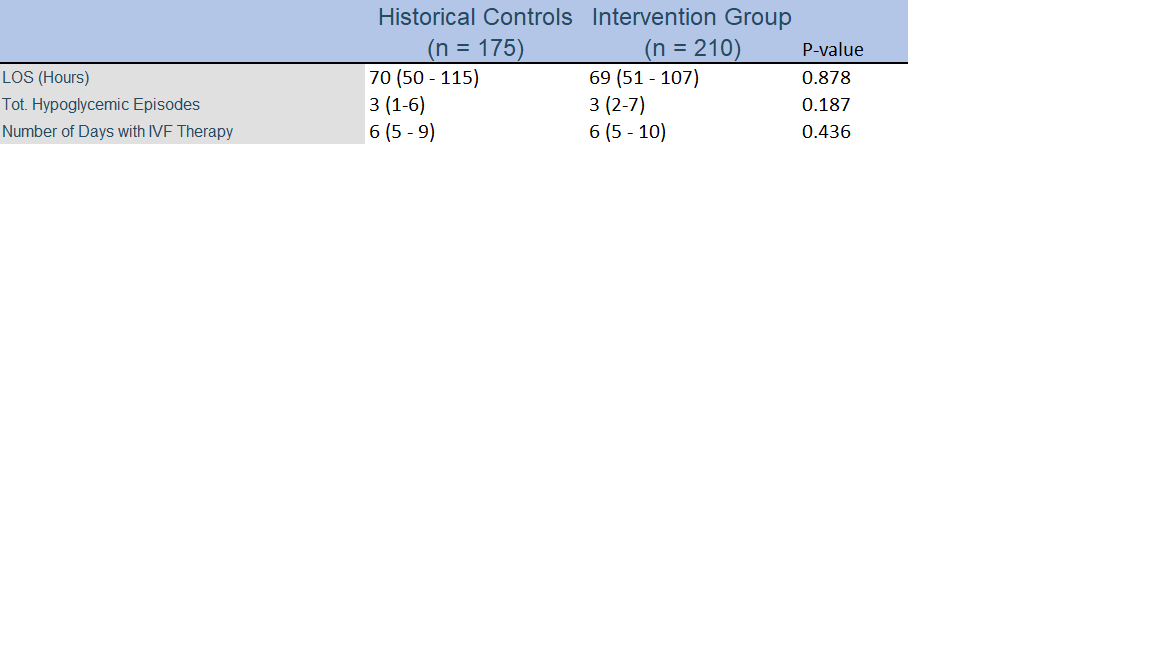 Outcomes of Intervention. All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
Outcomes of Intervention. All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
Objective: We evaluated the effects of introducing a clinical pathway for improving hypoglycemia with oral dextrose gel. We hypothesized a decrease in the length of stay of neonates with hypoglycemia after the introduction of the clinical pathway.
Design/Methods: We conducted a retrospective chart review evaluating neonates born ≥ 35 weeks gestational age, who exhibited asymptomatic hypoglycemia within 48 hours of life (HOL) while receiving exclusive oral feeds. The primary outcome was overall length of hospital stay. We extracted data from charts of children born prior and after the implementation of this oral dextrose gel pathway in both the well-baby nursery and Neonatal Intensive Care Unit (NICU) of a single medical center over a one-year period.
Results: 385 total neonates who fit the aforementioned criteria were included, with 175 neonates representing the historical control group and 210 neonates in the intervention group receiving oral dextrose gel. Analysis demonstrated no significant difference in the overall length of hospital stay, as well as the total episodes of hypoglycemia and number of days the neonate received intravenous fluid therapy with dextrose.Conclusion(s): Our study demonstrates that the introduction of oral dextrose gel pathway did not have a significant effect on the overall length of hospital stay, as well as the total episodes of hypoglycemia and number of days the neonate received intravenous fluid therapy with dextrose at our institution. Further research is needed to evaluate whether response to oral dextrose gel can be used as a predictor of severe hypoglycemia in the neonatal period.
Table 1. Characteristics of Study Subjects
.jpg) All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis TestTable 2. Results
 Outcomes of Intervention. All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test
Outcomes of Intervention. All categorical variables summarized with frequency and percentage, and compared across groups using the Fisher-Exact test. All numeric variables were summarized with median and 25th-75th percentile, and compared across groups using the Kruskal-Wallis Test