Back
Hospital Medicine: Systems/Population-based Research
Category: Abstract Submission
Hospital Medicine: Clinical NOS
321 - Characteristics Associated with Overuse of Reflux Medications in Infants
Saturday, April 23, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 321
Publication Number: 321.214
Publication Number: 321.214
Elizabeth R. Wolf, Children's Hospital of Richmond at VCU, Richmond, VA, United States; Martin Lavallee, Virginia Commonwealth University, Richmond, VA, United States; Roy T. Sabo, Virginia Commonwealth University School of Medicine, Richmond, VA, United States; Alan Schroeder, Stanford, Palo Alto, CA, United States; Alison N. Huffstetler, Virginia Commonwealth University School of Medicine, Alexandria, VA, United States; Matthew Schefft, Children's Hospital of Richmond at VCU, Richmond, VA, United States; Alex Krist, Virginia Commonwealth University School of Medicine, Fairfax, VA, United States
- EW
Elizabeth R. Wolf, MD, MPH
Assistant Professor
Children's Hospital of Richmond at VCU
Richmond, Virginia, United States
Presenting Author(s)
Background: The American Academy of Pediatrics and Choosing Wisely recommend against pharmacologic treatment of infant reflux because antacid medications are ineffective and may increase the risk for fractures and infections. Despite these recommendations, treatment of infant reflux with antacids is common. Little is known about the patient, provider and healthcare system characteristics associated with antacid overuse.
Objective: To determine the individual and healthcare system characteristics associated with antacid overuse using a statewide claims database.
Design/Methods: We used the Virginia All-Payer Claims Database from 2016-2019 to extract demographic data (gender, race, insurance type, zip code) and pharmacy claims for histamine type 2 receptor blockers (H2Bs) and proton pump inhibitors (PPIs) for children under 1 year of age. Children with a serious gastrointestinal disease, excessive weight loss, or medical complexity as defined by the Pediatric Complex Chronic Conditions classifications system and fewer than 30-days of continuous observation in the study period were excluded. Patient rurality was determined using the Education Demographic and Geographic Estimates (EDGE) Program from the National Center for Education Statistics. For the overuse outcome measure (prescription of H2B or PPI), we modeled the rate of the overuse using a binomial regression model, including gender, insurance type, patient rurality, and indicators for low birthweight ( < 2500 g) and prematurity ( < 37 weeks gestation). Race was excluded from the model because of the high degree of missingness.
Results: We identified 239,473 patients who met inclusion criteria of whom 5433 (2%) were prescribed a H2B or PPI. Children from rural settings had higher odds of being prescribed H2B/PPI compared to children from both urban/city (adjusted odds ratio [aOR] 1.23; 95% CI 1.12, 1.33) and suburban/town (aOR 1.60; 95% CI 1.48, 1.73) settings. Males tended to be prescribed a H2B/PPI slightly more often than females (aOR 1.11; 95% CI 1.05, 1.18). The odds of being prescribed H2B/PPI were higher (1.86; 95% CI 1.76, 1.97) for those with commercial insurance compared to public insurance. There were no significant differences in prescribing patterns for low-birthweight or premature infants.Conclusion(s): This study shows that overuse of antacid medications is more common in commercially insured infants and those living in rural locations. More research is needed to understand what drives overuse in these populations and what are the most effective tools to reduce unnecessary medications in children.
Table 1. Summary statistics for the population of children 0-1 years without a chronic condition or one of the medical exclusion criteria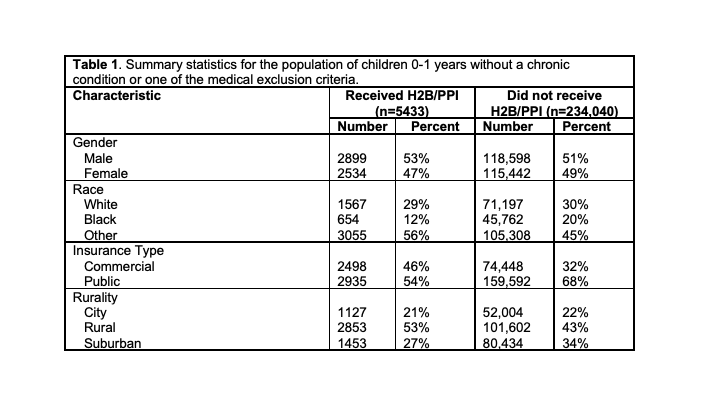
Table 2. Adjusted odds ratios and confidence intervals for prescription of Histamine type 2 Receptor Blocker (H2B) or Proton Pump Inhibitor (PPI)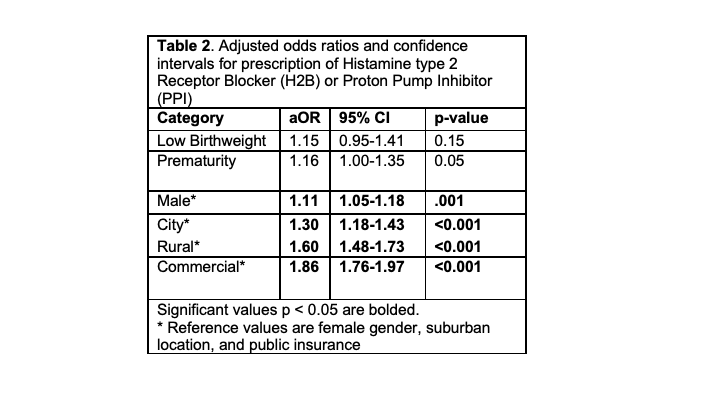
Objective: To determine the individual and healthcare system characteristics associated with antacid overuse using a statewide claims database.
Design/Methods: We used the Virginia All-Payer Claims Database from 2016-2019 to extract demographic data (gender, race, insurance type, zip code) and pharmacy claims for histamine type 2 receptor blockers (H2Bs) and proton pump inhibitors (PPIs) for children under 1 year of age. Children with a serious gastrointestinal disease, excessive weight loss, or medical complexity as defined by the Pediatric Complex Chronic Conditions classifications system and fewer than 30-days of continuous observation in the study period were excluded. Patient rurality was determined using the Education Demographic and Geographic Estimates (EDGE) Program from the National Center for Education Statistics. For the overuse outcome measure (prescription of H2B or PPI), we modeled the rate of the overuse using a binomial regression model, including gender, insurance type, patient rurality, and indicators for low birthweight ( < 2500 g) and prematurity ( < 37 weeks gestation). Race was excluded from the model because of the high degree of missingness.
Results: We identified 239,473 patients who met inclusion criteria of whom 5433 (2%) were prescribed a H2B or PPI. Children from rural settings had higher odds of being prescribed H2B/PPI compared to children from both urban/city (adjusted odds ratio [aOR] 1.23; 95% CI 1.12, 1.33) and suburban/town (aOR 1.60; 95% CI 1.48, 1.73) settings. Males tended to be prescribed a H2B/PPI slightly more often than females (aOR 1.11; 95% CI 1.05, 1.18). The odds of being prescribed H2B/PPI were higher (1.86; 95% CI 1.76, 1.97) for those with commercial insurance compared to public insurance. There were no significant differences in prescribing patterns for low-birthweight or premature infants.Conclusion(s): This study shows that overuse of antacid medications is more common in commercially insured infants and those living in rural locations. More research is needed to understand what drives overuse in these populations and what are the most effective tools to reduce unnecessary medications in children.
Table 1. Summary statistics for the population of children 0-1 years without a chronic condition or one of the medical exclusion criteria

Table 2. Adjusted odds ratios and confidence intervals for prescription of Histamine type 2 Receptor Blocker (H2B) or Proton Pump Inhibitor (PPI)

