Back
Pulmonology
Category: Abstract Submission
Pulmonology I
534 - Clinical Manifestations of E-cigarette, or Vaping Product Use-Associated Lung Injury (EVALI) in Adolescents Before and During the COVID-19 Pandemic
Saturday, April 23, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 534
Publication Number: 534.242
Publication Number: 534.242
Harin Lee, University of Texas Southwestern Medical School, Dallas, TX, United States; Bayan Mustafa N. Abdallah, University of Texas Southwestern Medical School, Irving, TX, United States; Sitara Weerakoon, University of Texas Health Science Center, Dallas, TX, United States; Sarah E. Messiah, University of Texas Health Science Center, School of Public Health, Dallas, TX, United States; Devika R. Rao, University of Texas Southwestern Medical School, Dallas, TX, United States
- HL
Harin Lee
Medical Student
University of Texas Southwestern Medical School
Dallas, Texas, United States
Presenting Author(s)
Background:
E-cigarette, or vaping, product use-associated lung injury (EVALI) results from inhaling the aerosol of e-cigarettes and has similar clinical features to coronavirus disease 2019 (COVID-19). EVALI case counts since the declaration of the COVID-19 pandemic is unknown.
Objective:
In this study, our aim was to determine how frequently COVID-19 was diagnosed in adolescents admitted to the hospital with EVALI during the COVID-19 pandemic. To account for a possible overlap in clinical symptoms between EVALI and COVID-19, with one affecting the severity of the other, our secondary aim was to compare clinical characteristics between two cohorts of EVALI patients, or those admitted: 1) prior to the pandemic; or 2) during the pandemic.
Design/Methods:
An electronic health record review of adolescents hospitalized at one institution with EVALI was conducted. Clinical characteristics and hospital course of patients hospitalized during the pandemic (DP) were compared to those pre-pandemic (PP).
Results:
A total of 41 adolescents diagnosed with confirmed/probable EVALI were included in the analysis (22= DP, 19=PP, Mean age 16.3 years, 66% male, 60% Hispanic). The clinical presentation of DP and PP adolescents were similar with respect to constitutional, respiratory and gastrointestinal symptoms. All DP patients were tested for COVID-19 at least once. Only one patient had a positive Severe Acute Respiratory Syndrome Coronavirus disease 2 Real-Time Polymerase Chain Reaction (SARS-CoV-2 RT-PCR) test result. 31 out of 39 patients treated with corticosteroids had clinical improvement within 24 hours (79%). DP patients had a shorter median length of stay (7 vs 5 days, p < 0.01), and were less often discharged with home oxygen (6 vs 1, p=0.04). PFTs for the overall cohort improved pre-to post-corticosteroid treatment (FEV1%, FVC%, 6-minute walk test distance and oxygen saturation) and post-corticosteroid to follow-up.
Conclusion(s):
Coexistent EVALI and COVID infection was uncommon during the pandemic. A shorter length of stay with less need for home oxygen in DP patients may reflect increased familiarity with EVALI characteristics. Corticosteroids led to clinical and pulmonary function improvement.
Harin Lee CVPAS H Lee CV.pdf
Pulmonary function test results pre-corticosteroid treatment, post-corticosteroid treatment, and on follow-up at the pulmonary clinic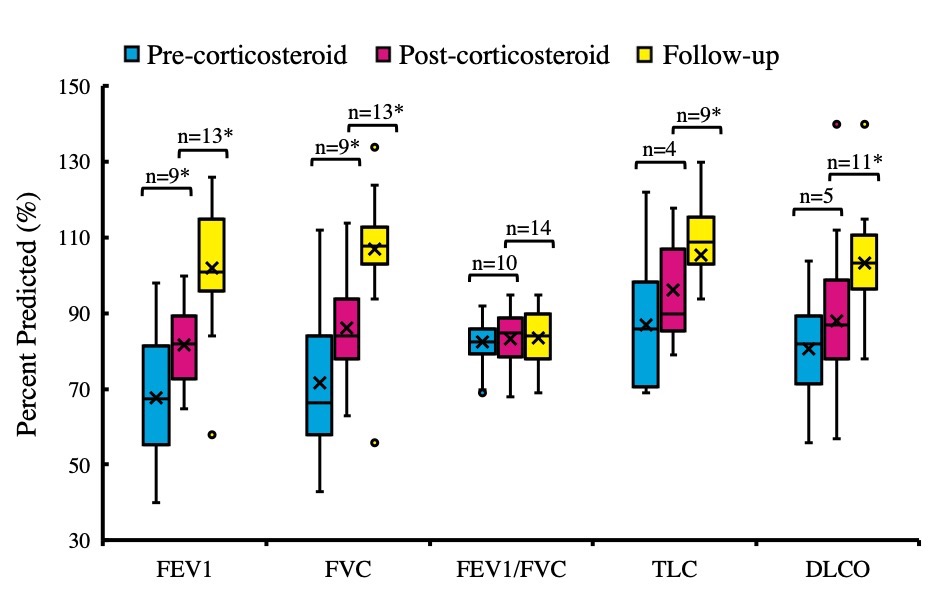 * = p < 0.05; ** = FEV1/FVC is an absolute ratio, not percent predicted
* = p < 0.05; ** = FEV1/FVC is an absolute ratio, not percent predicted
E-cigarette, or vaping, product use-associated lung injury (EVALI) results from inhaling the aerosol of e-cigarettes and has similar clinical features to coronavirus disease 2019 (COVID-19). EVALI case counts since the declaration of the COVID-19 pandemic is unknown.
Objective:
In this study, our aim was to determine how frequently COVID-19 was diagnosed in adolescents admitted to the hospital with EVALI during the COVID-19 pandemic. To account for a possible overlap in clinical symptoms between EVALI and COVID-19, with one affecting the severity of the other, our secondary aim was to compare clinical characteristics between two cohorts of EVALI patients, or those admitted: 1) prior to the pandemic; or 2) during the pandemic.
Design/Methods:
An electronic health record review of adolescents hospitalized at one institution with EVALI was conducted. Clinical characteristics and hospital course of patients hospitalized during the pandemic (DP) were compared to those pre-pandemic (PP).
Results:
A total of 41 adolescents diagnosed with confirmed/probable EVALI were included in the analysis (22= DP, 19=PP, Mean age 16.3 years, 66% male, 60% Hispanic). The clinical presentation of DP and PP adolescents were similar with respect to constitutional, respiratory and gastrointestinal symptoms. All DP patients were tested for COVID-19 at least once. Only one patient had a positive Severe Acute Respiratory Syndrome Coronavirus disease 2 Real-Time Polymerase Chain Reaction (SARS-CoV-2 RT-PCR) test result. 31 out of 39 patients treated with corticosteroids had clinical improvement within 24 hours (79%). DP patients had a shorter median length of stay (7 vs 5 days, p < 0.01), and were less often discharged with home oxygen (6 vs 1, p=0.04). PFTs for the overall cohort improved pre-to post-corticosteroid treatment (FEV1%, FVC%, 6-minute walk test distance and oxygen saturation) and post-corticosteroid to follow-up.
Conclusion(s):
Coexistent EVALI and COVID infection was uncommon during the pandemic. A shorter length of stay with less need for home oxygen in DP patients may reflect increased familiarity with EVALI characteristics. Corticosteroids led to clinical and pulmonary function improvement.
Harin Lee CVPAS H Lee CV.pdf
Pulmonary function test results pre-corticosteroid treatment, post-corticosteroid treatment, and on follow-up at the pulmonary clinic
 * = p < 0.05; ** = FEV1/FVC is an absolute ratio, not percent predicted
* = p < 0.05; ** = FEV1/FVC is an absolute ratio, not percent predicted