Back
Neonatal Pulmonology
Category: Abstract Submission
Neonatal Pulmonology V: Preclinical studies and Clinical Care Issues
483 - The SARS-CoV-2 E protein induces pro-inflammatory TLR signaling, lung injury and alveolar remodeling in the neonatal lung
Monday, April 25, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 483
Publication Number: 483.433
Publication Number: 483.433
Heather Menden, Children's Mercy Hospitals and Clinics, Kansas City, MO, United States; Wei Yu, Children's Mercy Kansas City, Kansas city, MO, United States; Sherry Mabry, Children's Mercy, Kansas City, MO, United States; Aparna Venkatraman, Children's Mercy Hospitals and Clinics, Kansas City, MO, United States; Todd Bradley, Children's Mercy Hospitals and Clinics, kansas city, MO, United States; Venkatesh Sampath, Childrens Mercy Kansas City, Kansas City, KS, United States
- HM
Heather Menden, MS
Lab Operations Manager, Research Associate MS
Childrens Mercy
Kansas City, Missouri, United States
Presenting Author(s)
Background: The delta variant of SARS-CoV-2 is reported to cause severe acute lung injury (ALI) in neonates. Neonates with COVID-19 can exhibit a hyper-inflammatory response with vascular injury. The mechanisms by which SARS-CoV-2 structural proteins induce ALI in neonates is unclear. Emerging data suggest that the envelope (E protein) of the SARS-CoV-2 activates Toll Like Receptor (TLR)-mediated immune hyperactivation and cytokine storm. We tested the hypothesis that SARS-CoV-2 E protein can induce TLR-mediated inflammation and ALI in a neonatal mouse model of ALI we developed.
Objective: 1) To investigate E protein pathogenicity in neonatal ALI and alveolar remodeling in an in vivo mouse model, 2) To determine whether E protein induces TLR-dependent cell death and inflammation in human pulmonary microvascular endothelial cells (HPMEC).
Design/Methods: We developed a model of systemic E protein-induced ALI using intraperitoneal (i.p.) injections of 10μg (2mg/kg) in P5 [saccular stage] C57BL6 mice. Lung homogenates were used for quantifying cytokine RNA expression and TLR signaling 48 hours (h) after E protein injections. Bronchoalveolar lavage (BAL) was done on P11 mice 48h after i.p E protein. E protein-induced alveolar remodeling was examined by morphometry on P14 after injecting E protein at P5. Neonatal HPMEC (ScienCell) were treated with 500ng/mL E protein, with or without silencing of TLR2 (siTLR2), and cell lysates were examined for cytokine expression and TLR signaling.
Results: E protein induced lung Cxcl1, iNOS, Il-1β, and Tnfα gene expression in a dose-dependent manner at 48h. Activation of TLR signaling was evidenced by phosphorylation of inhibitor of kappa B kinase β (pIKKβ) and the NFKB subunit p65 (Fig.1). BAL showed increased albumin content and neutrophil and lymphocyte infiltration 48h after i.p E protein. A single dose of E protein given on P5 (late saccular stage) induced alveolar simplification that persisted at P14 (mid-alveolar phase) as evidenced by decreased alveolar counts. (Fig. 2). In HPMEC, E protein induced HPMEC cell death in a dose-dependent manner, as well as TLR signaling [p-IKKβ], NFKB activation [p-p65], and MAPK signaling [p-p38]. E protein-induced ICAM1, IL-1β, TNFα, IL6, IL8, and TLR2 expression was suppressed by silencing TLR2. (Fig. 3)Conclusion(s): The SARS-CoV-2 E protein induced TLR signaling, ALI and alveolar remodeling in the neonatal mouse lung. Ongoing studies suggest that E protein induces direct EC injury in human lung EC that is TLR2-dependent. This study provides mechanistic insight into neonatal immune activation and lung injury seen in infants with COVID-19.
Figure 1. Lung inflammation and immune signaling induced in mice 48hr post E protein treatment.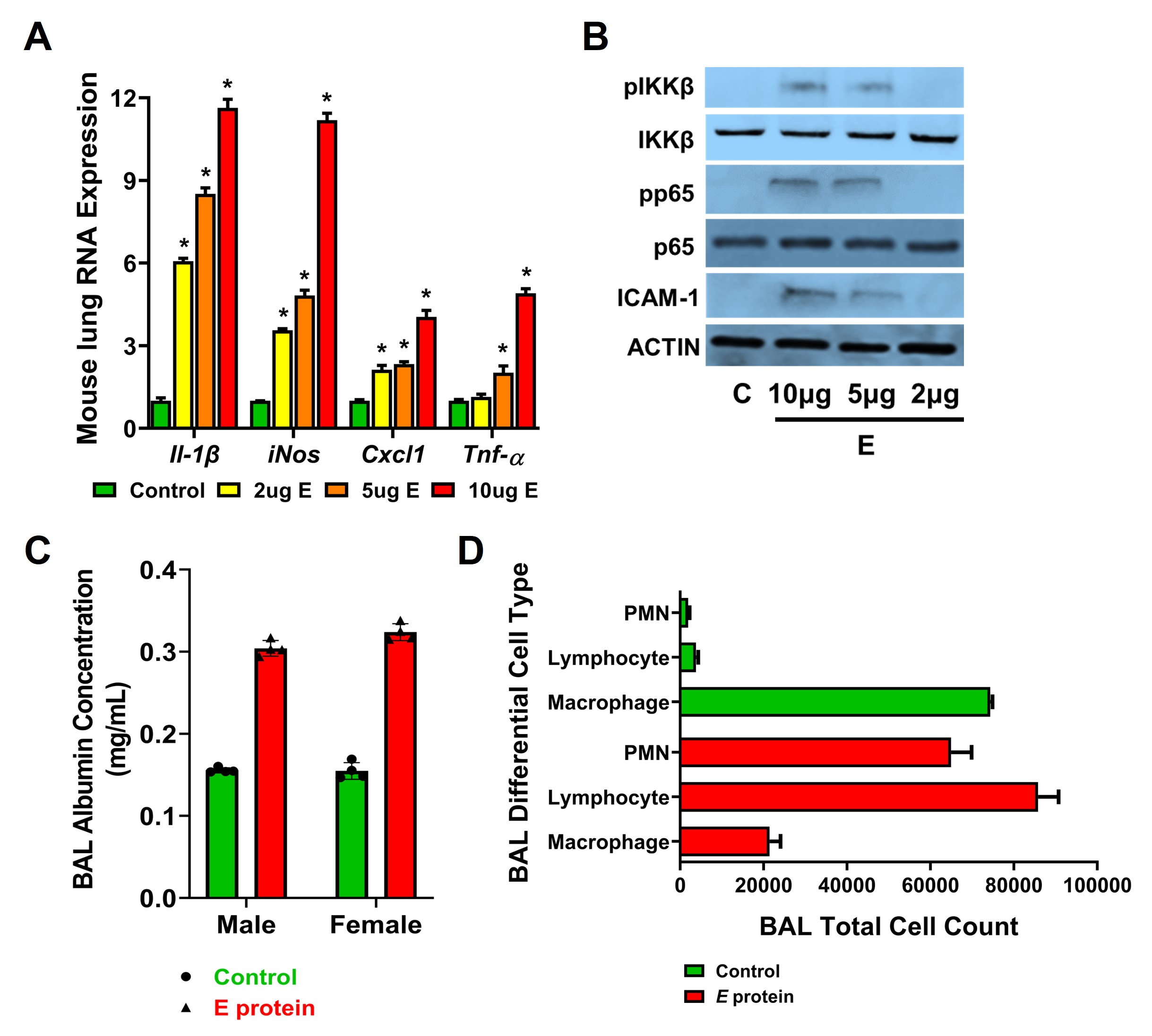 (A) qRT-PCR of genes associated with inflammation from the lung lysates of P7 mice after E protein injection at three doses (2µg (yellow), 5µg (orange), 10µg (red) or untreated control (green). (n=4 mice per group). *p < 0.01 (Control vs. E protein dose). (B) Western blot from lung lysates after TLR activation (phosphorylation of IKKβ and p65, pIKKβ, and pp65) and ICAM expression qualified. n=4. (C-D) Bronchoalveolar lung lavages (BAL) were done on P11 mice following E treatments, with the levels of albumin quantified (C) and the total cell counts with cellular differential shown (D). n=4 for each male and female. *p < 0.01 (Control vs. E protein).
(A) qRT-PCR of genes associated with inflammation from the lung lysates of P7 mice after E protein injection at three doses (2µg (yellow), 5µg (orange), 10µg (red) or untreated control (green). (n=4 mice per group). *p < 0.01 (Control vs. E protein dose). (B) Western blot from lung lysates after TLR activation (phosphorylation of IKKβ and p65, pIKKβ, and pp65) and ICAM expression qualified. n=4. (C-D) Bronchoalveolar lung lavages (BAL) were done on P11 mice following E treatments, with the levels of albumin quantified (C) and the total cell counts with cellular differential shown (D). n=4 for each male and female. *p < 0.01 (Control vs. E protein).
Objective: 1) To investigate E protein pathogenicity in neonatal ALI and alveolar remodeling in an in vivo mouse model, 2) To determine whether E protein induces TLR-dependent cell death and inflammation in human pulmonary microvascular endothelial cells (HPMEC).
Design/Methods: We developed a model of systemic E protein-induced ALI using intraperitoneal (i.p.) injections of 10μg (2mg/kg) in P5 [saccular stage] C57BL6 mice. Lung homogenates were used for quantifying cytokine RNA expression and TLR signaling 48 hours (h) after E protein injections. Bronchoalveolar lavage (BAL) was done on P11 mice 48h after i.p E protein. E protein-induced alveolar remodeling was examined by morphometry on P14 after injecting E protein at P5. Neonatal HPMEC (ScienCell) were treated with 500ng/mL E protein, with or without silencing of TLR2 (siTLR2), and cell lysates were examined for cytokine expression and TLR signaling.
Results: E protein induced lung Cxcl1, iNOS, Il-1β, and Tnfα gene expression in a dose-dependent manner at 48h. Activation of TLR signaling was evidenced by phosphorylation of inhibitor of kappa B kinase β (pIKKβ) and the NFKB subunit p65 (Fig.1). BAL showed increased albumin content and neutrophil and lymphocyte infiltration 48h after i.p E protein. A single dose of E protein given on P5 (late saccular stage) induced alveolar simplification that persisted at P14 (mid-alveolar phase) as evidenced by decreased alveolar counts. (Fig. 2). In HPMEC, E protein induced HPMEC cell death in a dose-dependent manner, as well as TLR signaling [p-IKKβ], NFKB activation [p-p65], and MAPK signaling [p-p38]. E protein-induced ICAM1, IL-1β, TNFα, IL6, IL8, and TLR2 expression was suppressed by silencing TLR2. (Fig. 3)Conclusion(s): The SARS-CoV-2 E protein induced TLR signaling, ALI and alveolar remodeling in the neonatal mouse lung. Ongoing studies suggest that E protein induces direct EC injury in human lung EC that is TLR2-dependent. This study provides mechanistic insight into neonatal immune activation and lung injury seen in infants with COVID-19.
Figure 1. Lung inflammation and immune signaling induced in mice 48hr post E protein treatment.
 (A) qRT-PCR of genes associated with inflammation from the lung lysates of P7 mice after E protein injection at three doses (2µg (yellow), 5µg (orange), 10µg (red) or untreated control (green). (n=4 mice per group). *p < 0.01 (Control vs. E protein dose). (B) Western blot from lung lysates after TLR activation (phosphorylation of IKKβ and p65, pIKKβ, and pp65) and ICAM expression qualified. n=4. (C-D) Bronchoalveolar lung lavages (BAL) were done on P11 mice following E treatments, with the levels of albumin quantified (C) and the total cell counts with cellular differential shown (D). n=4 for each male and female. *p < 0.01 (Control vs. E protein).
(A) qRT-PCR of genes associated with inflammation from the lung lysates of P7 mice after E protein injection at three doses (2µg (yellow), 5µg (orange), 10µg (red) or untreated control (green). (n=4 mice per group). *p < 0.01 (Control vs. E protein dose). (B) Western blot from lung lysates after TLR activation (phosphorylation of IKKβ and p65, pIKKβ, and pp65) and ICAM expression qualified. n=4. (C-D) Bronchoalveolar lung lavages (BAL) were done on P11 mice following E treatments, with the levels of albumin quantified (C) and the total cell counts with cellular differential shown (D). n=4 for each male and female. *p < 0.01 (Control vs. E protein).
Designed by Cadmium
|Technical Support
© Copyright 2024 Cadmium. All Rights Reserved.
