Back
Neonatal Pulmonology
Category: Abstract Submission
Neonatal Pulmonology III: Molecular Markers and Clinical Prediction Models/Outcomes
454 - Prediction models for bronchopulmonary dysplasia in preterm children: a systematic review and meta-analysis
Monday, April 25, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 454
Publication Number: 454.431
Publication Number: 454.431
Michelle Romijn, Emma Children's Hospital Amsterdam UMC, Amsterdam, Noord-Holland, Netherlands; Paula Dhiman, University of Oxford, Oxford, England, United Kingdom; Martijn J. Finken, Emma Children's Hospital, Amsterdam, Noord-Holland, Netherlands; Anton van Kaam, Emma Children's Hospitial, Amsterdam UMC, Amsterdam, Noord-Holland, Netherlands; Trix A. Katz, Amsterdam University Medical Center, location AMC, Amsterdam, Noord-Holland, Netherlands; Joost Rotteveel, Amsterdam UMC, Amsterdam, Noord-Holland, Netherlands; Ewoud Schuit, University Medical Center Utrecht, Utrecht, Utrecht, Netherlands; Gary Collins, University of Oxford, Oxford, England, United Kingdom; Wes Onland, Emma Children's Hospital Amsterdam UMC, Amsterdam, Noord-Holland, Netherlands; Heloise Torchin, INSERM, APHP, Paris, Ile-de-France, France

Michelle Romijn, MD
Research Fellow
Emma Children's Hospital Amsterdam UMC
Amsterdam, Noord-Holland, Netherlands
Presenting Author(s)
Background: Bronchopulmonary dysplasia (BPD) remains a frequent complication of preterm birth, affecting more than one third of the infants born before 28 weeks of gestation. Early identification of infants developing BPD is of the utmost importance for increased precision of treating infants with a high-risk profile. A 2013 systematic review identified 26 BPD prediction models using routinely collected clinical data during the first week of life. Since then, many new prediction models have been published, including models investigating potential biomarkers. Furthermore, the Prediction model Risk Of Bias Assessment Tool (PROBAST) has since been published to provide a formal Risk of Bias assessment for studies aiming to develop or validate clinical prediction models.
Objective: To systematically review multivariable prediction models for BPD or BPD/death at 36 weeks postmenstrual age.
Design/Methods: The search and identification of eligible studies were performed by using the electronic databases MEDLINE and EMBASE. All studies published between 1990-2020 developing or validating a multivariable prediction model for BPD or BPD/death in the first 14 days of life were included. Data were extracted by two independent authors, according to the CHecklist for critical Appraisal and data extraction for systematic Reviews of prediction Modelling Studies. Risk of bias was assessed using PROBAST. Meta-analysis was performed combining all published studies with external validation on day 1, 3, 7 and 14 in order to investigate predictive performance on different time points.
Results: Forty-four studies were included, containing 53 developed and 64 validated models. A median c-statistic of 0.84 [min-max 0.72-1.00] was found for the development models, and 0.81 [min-max 0.60-0.97] for the validated models. Following PROBAST, all models had a high risk of bias due to small sample sizes, inadequate management or reporting of missing data, absence or inadequate evaluation of calibration performances. Meta-analysis of the validated models revealed that c-statistics increased from day 1 to day 7 for the outcome BPD, but only from day 1 to day 3 for the outcome BPD/death (see Figure 1a,b).Conclusion(s): Although the BPD prediction models perform quite well, the models need methodological improvement and more accurate reporting before they can be used in clinical practice. Future research should aim to validate and update already existing models strictly following the PROBAST guideline, and investigate if dynamic prediction models would improve discriminating performances at different time points.
Figure 1a. Meta-analysis of validated models on day 1, 3, 7 and 14 with outcome BPD.png)
Figure 1b. Meta-analysis of validated models on day 1, 3, 7 and 14 with outcome BPD or Death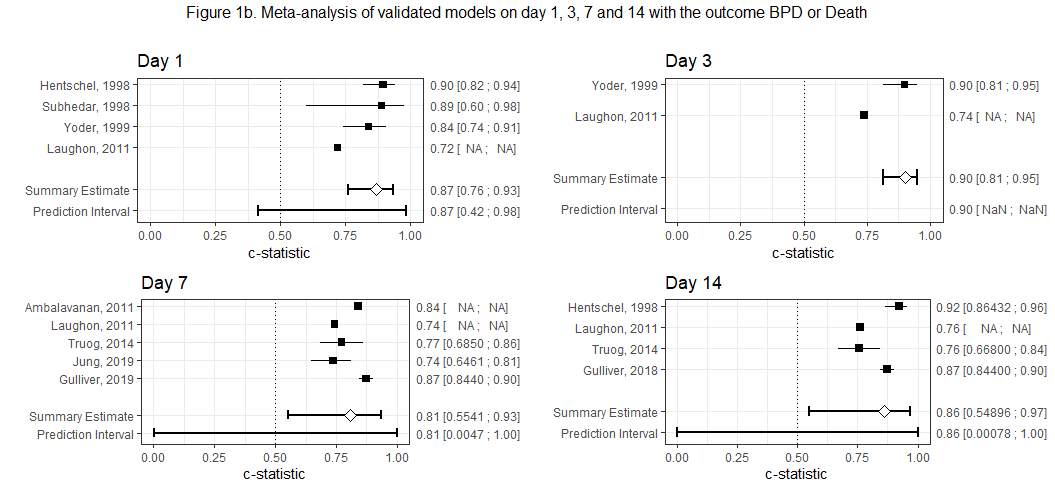
Objective: To systematically review multivariable prediction models for BPD or BPD/death at 36 weeks postmenstrual age.
Design/Methods: The search and identification of eligible studies were performed by using the electronic databases MEDLINE and EMBASE. All studies published between 1990-2020 developing or validating a multivariable prediction model for BPD or BPD/death in the first 14 days of life were included. Data were extracted by two independent authors, according to the CHecklist for critical Appraisal and data extraction for systematic Reviews of prediction Modelling Studies. Risk of bias was assessed using PROBAST. Meta-analysis was performed combining all published studies with external validation on day 1, 3, 7 and 14 in order to investigate predictive performance on different time points.
Results: Forty-four studies were included, containing 53 developed and 64 validated models. A median c-statistic of 0.84 [min-max 0.72-1.00] was found for the development models, and 0.81 [min-max 0.60-0.97] for the validated models. Following PROBAST, all models had a high risk of bias due to small sample sizes, inadequate management or reporting of missing data, absence or inadequate evaluation of calibration performances. Meta-analysis of the validated models revealed that c-statistics increased from day 1 to day 7 for the outcome BPD, but only from day 1 to day 3 for the outcome BPD/death (see Figure 1a,b).Conclusion(s): Although the BPD prediction models perform quite well, the models need methodological improvement and more accurate reporting before they can be used in clinical practice. Future research should aim to validate and update already existing models strictly following the PROBAST guideline, and investigate if dynamic prediction models would improve discriminating performances at different time points.
Figure 1a. Meta-analysis of validated models on day 1, 3, 7 and 14 with outcome BPD
.png)
Figure 1b. Meta-analysis of validated models on day 1, 3, 7 and 14 with outcome BPD or Death

