Back
Neonatal Pulmonology
Category: Abstract Submission
Neonatal Pulmonology II: Therapies and Emerging Therapies for BPD
440 - Safety, pharmacokinetic, and mechanistic studies of AVR-48 in the preterm lamb model of bronchopulmonary dysplasia
Monday, April 25, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 440
Publication Number: 440.430
Publication Number: 440.430
Suchismita Acharya, AyuVis Research Inc, Fort Worth, TX, United States; Dale J. Christensen, Duke University School of Medicine, Cary, NC, United States; David Riley, Texas Christian University and UNT Health Science Center School of Medicine, Westlake, TX, United States; Sumita Behera, Ayuvis research, Flowermound, TX, United States; Andrew Rebentisch, University of Utah - division of neonatology, Salt Lake City, UT, United States; Elaine Dawson, University of Utah, Newport Beach, CA, United States; Emily Major, University of Utah, Salt Lake City, UT, United States; Jakob D. Van Boerum, University of Utah, Holladay, UT, United States; Eesha Acharya, Ayuvis Research Inc, Grapevine, TX, United States; Kurt H. Albertine, University of Utah School of Medicine, Salt Lake City, UT, United States

Suchismita Acharya, PhD
Chief Executive and Chief Scientific Officer
AyuVis Research Inc
Fort Worth, Texas, United States
Presenting Author(s)
Background: AVR-48 is a novel immunomodulatory molecule derived from chitin and previously shown safe and effective in preventing hyperoxia-induced pathologies in a mouse pup model of bronchopulmonary dysplasia (BPD).
Objective: To demonstrate safety of AVR-48 in a large-animal model that mimics preterm infants developing BPD in the NICU setting and determine pharmacokinetic parameters following intravenous (IV) dosing twice daily for 7days.
Design/Methods: Preterm lambs delivered at ~128 days gestation post-antenatal steroids were intubated, given surfactant and managed by invasive mechanical ventilation (IMV) for 7d followed by 3d of noninvasive respiratory support (NRS). Either placebo (saline) or AVR-48 (1.0 or 3.0 mg/kg) was given IV q12h for 7d, beginning 6h after delivery (n=2-3 lambs per dose). Blood samples were collected at 19 specified time points for PK study, clinical biochemistry (ARUP lab), and cytokine assays (ELISA). Lung homogenates were analyzed for cleaved caspase 3, PCNA, VEGF, VEGF-R2, and other biomarkers, using both immunoblot and RT-PCR.
Results: Fig 1: Maximum drug concentration (Cmax) of AVR-48 (3.0 mg/kg/dose; efficacy dose) in plasma after the first dose showed linear decline in drug concentration, with half-life ~30 min and Cmax= 10.73 µM (Fig 1A; for reference, Cmax of AVR-48 in mouse pups at efficacy dose of 10 mg/kg IP injection was 5.7 µM). The half-life of AVR-48 (~4h) in PT lambs is like juvenile rats after repeat dosing (2/d for 7d), with no drug accumulation at 7d (Fig 1B). Fig 2: AVR-48-treated PT lambs (3.0 mg/kg, n=3) had normal plasma clinical biochemistry relative to control PT lambs. Fig 3: Lower plasma level of the inflammatory cytokine IL-1β (Fig 3A) in AVR-48-treated PT lambs were observed, with higher level of the anti-inflammatory cytokine IL-10 (Fig 3B), relative to control PT lambs. Fig 3C indicates greater protein abundance of cleaved caspase 3, and lower protein abundance of PCNA, together which are associated with appropriate thinning of septal walls to alveolar walls in lung. Both immunoblot (Fig 3C) and RT-PCR (Fig 3D) of lung homogenates indicate greater mRNA level of VEGF-R2 after AVR-48 treatment.Conclusion(s): PK of AVR-48 demonstrated linear elimination with consistent and predictable plasma levels across a wide dosing range. No significant renal or hepatic toxicity is observed. Inflammatory cytokine markers measured in PT lamb plasma confirm a mechanism of action consistent with our previous in vitro and rodent models.
Figure 1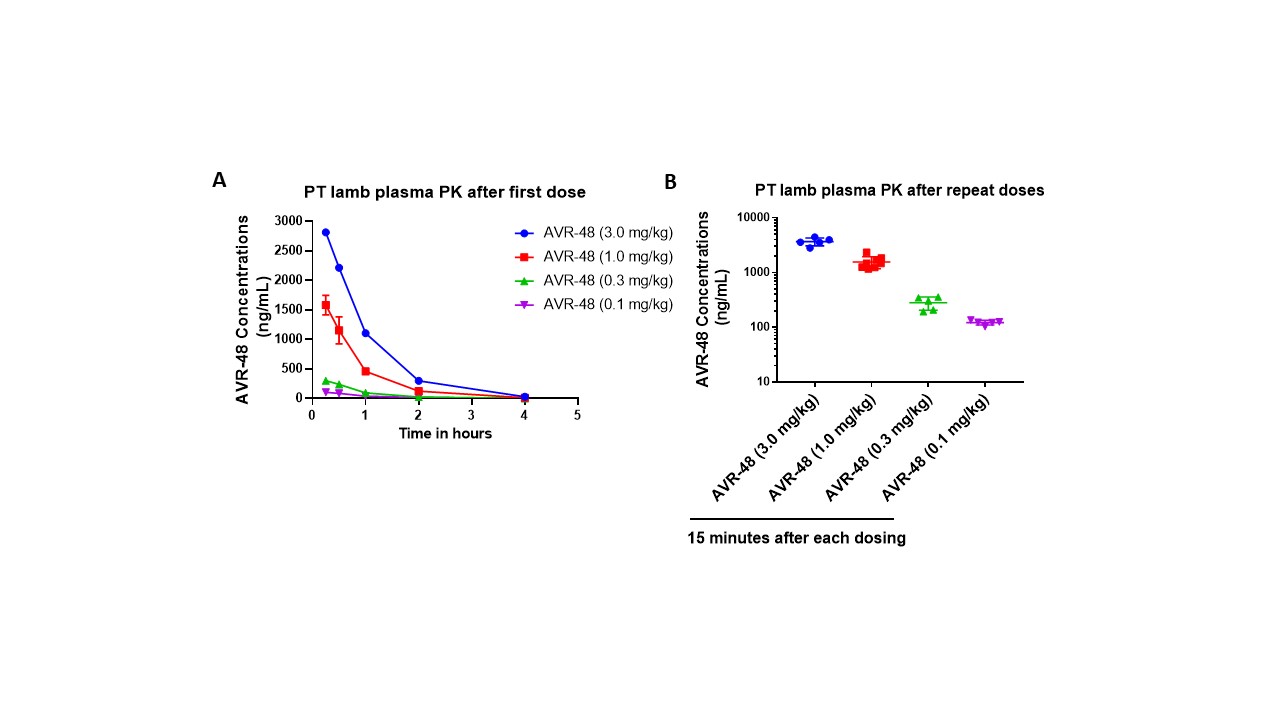 Preterm lamb plasma PK data for AVR-48 via intravenous dosing. A) after first dose. B) After repeat doses
Preterm lamb plasma PK data for AVR-48 via intravenous dosing. A) after first dose. B) After repeat doses
Figure 2.jpg) Prophylactic treatment of AVR-48 did not cause acute liver or kidney injury in PT lambs (End-of-study plasma clinical biochemistry profiles)
Prophylactic treatment of AVR-48 did not cause acute liver or kidney injury in PT lambs (End-of-study plasma clinical biochemistry profiles)
Objective: To demonstrate safety of AVR-48 in a large-animal model that mimics preterm infants developing BPD in the NICU setting and determine pharmacokinetic parameters following intravenous (IV) dosing twice daily for 7days.
Design/Methods: Preterm lambs delivered at ~128 days gestation post-antenatal steroids were intubated, given surfactant and managed by invasive mechanical ventilation (IMV) for 7d followed by 3d of noninvasive respiratory support (NRS). Either placebo (saline) or AVR-48 (1.0 or 3.0 mg/kg) was given IV q12h for 7d, beginning 6h after delivery (n=2-3 lambs per dose). Blood samples were collected at 19 specified time points for PK study, clinical biochemistry (ARUP lab), and cytokine assays (ELISA). Lung homogenates were analyzed for cleaved caspase 3, PCNA, VEGF, VEGF-R2, and other biomarkers, using both immunoblot and RT-PCR.
Results: Fig 1: Maximum drug concentration (Cmax) of AVR-48 (3.0 mg/kg/dose; efficacy dose) in plasma after the first dose showed linear decline in drug concentration, with half-life ~30 min and Cmax= 10.73 µM (Fig 1A; for reference, Cmax of AVR-48 in mouse pups at efficacy dose of 10 mg/kg IP injection was 5.7 µM). The half-life of AVR-48 (~4h) in PT lambs is like juvenile rats after repeat dosing (2/d for 7d), with no drug accumulation at 7d (Fig 1B). Fig 2: AVR-48-treated PT lambs (3.0 mg/kg, n=3) had normal plasma clinical biochemistry relative to control PT lambs. Fig 3: Lower plasma level of the inflammatory cytokine IL-1β (Fig 3A) in AVR-48-treated PT lambs were observed, with higher level of the anti-inflammatory cytokine IL-10 (Fig 3B), relative to control PT lambs. Fig 3C indicates greater protein abundance of cleaved caspase 3, and lower protein abundance of PCNA, together which are associated with appropriate thinning of septal walls to alveolar walls in lung. Both immunoblot (Fig 3C) and RT-PCR (Fig 3D) of lung homogenates indicate greater mRNA level of VEGF-R2 after AVR-48 treatment.Conclusion(s): PK of AVR-48 demonstrated linear elimination with consistent and predictable plasma levels across a wide dosing range. No significant renal or hepatic toxicity is observed. Inflammatory cytokine markers measured in PT lamb plasma confirm a mechanism of action consistent with our previous in vitro and rodent models.
Figure 1
 Preterm lamb plasma PK data for AVR-48 via intravenous dosing. A) after first dose. B) After repeat doses
Preterm lamb plasma PK data for AVR-48 via intravenous dosing. A) after first dose. B) After repeat dosesFigure 2
.jpg) Prophylactic treatment of AVR-48 did not cause acute liver or kidney injury in PT lambs (End-of-study plasma clinical biochemistry profiles)
Prophylactic treatment of AVR-48 did not cause acute liver or kidney injury in PT lambs (End-of-study plasma clinical biochemistry profiles)