Back
Neonatal Infectious Diseases/Immunology
Category: Abstract Submission
Neonatal Infectious Diseases/Immunology: Sepsis
561 - Biomarkers to Estimate Baseline Risk of Mortality in Neonates with Sepsis and Necrotizing Enterocolitis
Monday, April 25, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 561
Publication Number: 561.427
Publication Number: 561.427
Faris N. Al Gharaibeh, Cincinnati Children's Hospital Medical Center, cincinnati, OH, United States; Patrick Lahni, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States; Matthew N. Alder, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States; Hector Wong, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States
.jpg)
Faris N. Al Gharaibeh, MD
Clinical Fellow
Cincinnati Children's Hospital Medical Center
cincinnati, Ohio, United States
Presenting Author(s)
Background: Biomarker research in sepsis has been heavily focused on identifying culture-positive sepsis. Biomarkers can also have utility for estimation of baseline mortality risk, which is fundamental to clinical practice and research. PERSEVERE is a multi-biomarker (CCL3, granzyme B, HSP-70, IL-8, MMP-8, and platelet count) decision tree that is now validated to reliably estimate baseline risk of mortality among children with septic shock.
Objective: Test if PERSEVERE can reliably estimate baseline risk of mortality among neonates with sepsis or necrotizing enterocolitis. If PERSEVERE did not perform well, we planned to derive a neonatal specific decision tree using PERSEVERE biomarkers.
Design/Methods: This is a prospective cohort of neonates admitted to Cincinnati Children’s Hospital Medical Center NICU and the University of Cincinnati Medical Center NICU. Patients were enrolled if they met criteria for sepsis according to Wynn et al., 2011 or had NEC stage II or greater according to Bell et al., 1978. Neonates were excluded if they only received 48 hours of antibiotics or found to have a lethal diagnosis. Residual whole blood at the time of sepsis/NEC evaluation was obtained for biomarker analysis using Luminex technology. The primary outcome is mortality at discharge and the secondary outcome is complicated course of illness (defined as death or the occurrence of 2 or more organ dysfunction on day 7 of illness).
Results: 71 neonates had 64 sepsis event and 14 NEC events with a mortality rate of 18.3% (table 1). If a neonate had multiple events, we included only the last event in the analysis. PERSEVERE classified neonates into a higher risk group (mortality 29.4%, complicated course 41.8%) and lower risk group (mortality 8.1%, complicated course 16.2%). It was 77% sensitive and 59% specific for mortality, p 0.03 (table 2). We derived a new tree for neonates (nPERSEVERE) using Classification and Regression Tree methodology (Figure 1). nPERSEVERE had an AUROC of 0.89, with 92% sensitivity and 76% specificity for estimating the risk of mortality. Upon 10-fold cross validation the summary AUROC was 0.73. Among patients classified to terminal nodes 3 and 4, the mortality rate was 46% and the complicated course rate was 66%. In contrast, among patients classified to terminal nodes 1 and 2, the mortality rate was 2% and complicated course rate was 13%, p < 0.0001Conclusion(s): nPERSEVERE could have utility for identifying neonates at higher risk of dying from sepsis and NEC who could benefit from high-risk therapies and aid in clinical trial enrichment. Our findings need validation in a larger multi-center cohort.
Table 1. Characteristics of Neonates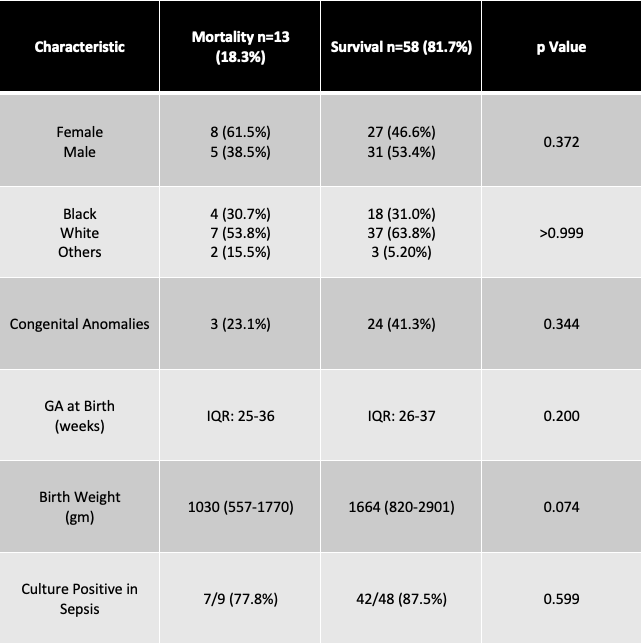 Neonates who experienced mortality at discharge were comparable to those who did not. There was a statistically insignificant trend for lower gestational age at birth and lesser weight in those who died compared to those who survived.
Neonates who experienced mortality at discharge were comparable to those who did not. There was a statistically insignificant trend for lower gestational age at birth and lesser weight in those who died compared to those who survived.
Table 2. PERSEVERE and nPERSEVERE Performance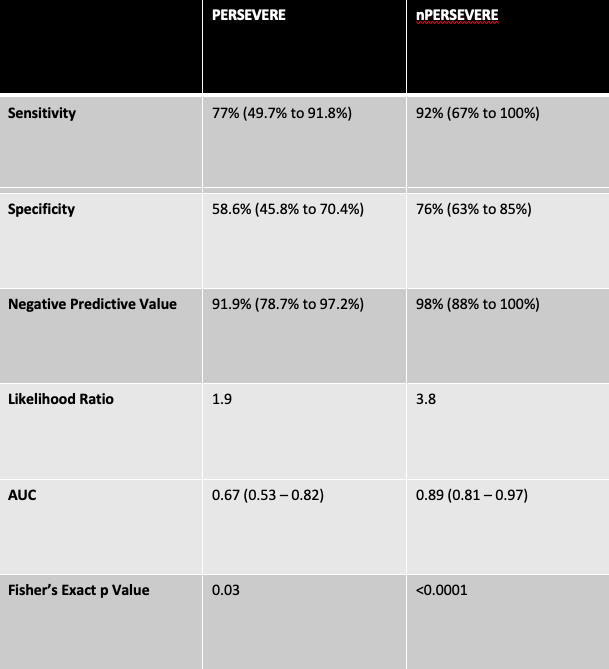
Objective: Test if PERSEVERE can reliably estimate baseline risk of mortality among neonates with sepsis or necrotizing enterocolitis. If PERSEVERE did not perform well, we planned to derive a neonatal specific decision tree using PERSEVERE biomarkers.
Design/Methods: This is a prospective cohort of neonates admitted to Cincinnati Children’s Hospital Medical Center NICU and the University of Cincinnati Medical Center NICU. Patients were enrolled if they met criteria for sepsis according to Wynn et al., 2011 or had NEC stage II or greater according to Bell et al., 1978. Neonates were excluded if they only received 48 hours of antibiotics or found to have a lethal diagnosis. Residual whole blood at the time of sepsis/NEC evaluation was obtained for biomarker analysis using Luminex technology. The primary outcome is mortality at discharge and the secondary outcome is complicated course of illness (defined as death or the occurrence of 2 or more organ dysfunction on day 7 of illness).
Results: 71 neonates had 64 sepsis event and 14 NEC events with a mortality rate of 18.3% (table 1). If a neonate had multiple events, we included only the last event in the analysis. PERSEVERE classified neonates into a higher risk group (mortality 29.4%, complicated course 41.8%) and lower risk group (mortality 8.1%, complicated course 16.2%). It was 77% sensitive and 59% specific for mortality, p 0.03 (table 2). We derived a new tree for neonates (nPERSEVERE) using Classification and Regression Tree methodology (Figure 1). nPERSEVERE had an AUROC of 0.89, with 92% sensitivity and 76% specificity for estimating the risk of mortality. Upon 10-fold cross validation the summary AUROC was 0.73. Among patients classified to terminal nodes 3 and 4, the mortality rate was 46% and the complicated course rate was 66%. In contrast, among patients classified to terminal nodes 1 and 2, the mortality rate was 2% and complicated course rate was 13%, p < 0.0001Conclusion(s): nPERSEVERE could have utility for identifying neonates at higher risk of dying from sepsis and NEC who could benefit from high-risk therapies and aid in clinical trial enrichment. Our findings need validation in a larger multi-center cohort.
Table 1. Characteristics of Neonates
 Neonates who experienced mortality at discharge were comparable to those who did not. There was a statistically insignificant trend for lower gestational age at birth and lesser weight in those who died compared to those who survived.
Neonates who experienced mortality at discharge were comparable to those who did not. There was a statistically insignificant trend for lower gestational age at birth and lesser weight in those who died compared to those who survived.Table 2. PERSEVERE and nPERSEVERE Performance

