Back
Nephrology: CKD
Category: Abstract Submission
Nephrology IV: Chronic Kidney Disease
59 - Anemia, Iron Deficiency and FGF23 in CKiD Study
Sunday, April 24, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 59
Publication Number: 59.341
Publication Number: 59.341
Elizabeth Thomas, UCLA Mattel Childrens Hospital, Culver City, CA, United States; Marciana Laster, UCLA Mattel Childrens Hospital, Los Angeles, CA, United States; Susan Furth, Childrens Hospital of Philadelphia, Philadelphia, PA, United States; BRADLEY A. WARADY, CHILDREN'S MERCY KANSAS CITY, KANSAS CITY, MO, United States; Alexandra M. Klomhaus, University of California, Los Angeles David Geffen School of Medicine, Los Angeles, CA, United States; Isidro B. Salusky, University of California, Los Angeles David Geffen School of Medicine, Los Angeles, CA, United States; Mark R. Hanudel, University of California, Los Angeles David Geffen School of Medicine, Los Angeles, CA, United States
- ET
Elizabeth Thomas, MD
Pediatric Nephrology Fellow
UCLA Mattel Childrens Hospital
Culver City, California, United States
Presenting Author(s)
Background: Fibroblast growth factor 23 (FGF23) is a bone-derived hormone fundamental to chronic kidney disease-mineral bone disorder (CKD-MBD), CKD progression, and CKD-associated cardiovascular morbidity. It has recently been demonstrated that non-mineral factors, such as iron deficiency and erythropoietin (EPO), can increase FGF23 production. CKD-associated anemia can be characterized by iron deficiency and/or compensatorily increased EPO levels.
Objective: The primary aims of this study are to assess whether anemia and/or iron deficiency are associated with increased FGF23 concentrations.
Design/Methods: Anemia, iron and FGF23 profiles were characterized in cross-sectional and longitudinal analyses of subjects enrolled in the CKiD study. Participants included children aged 1 month to 16 years old with mild to moderate CKD.
Results: In a cross-sectional analysis of 686 pediatric CKD patients at baseline (median (interquartile range) age 11 (8, 15) years, 62% male, 14% Hispanic), the median estimated glomerular filtration rate (eGFR) was 55 (41, 71) ml/min/1.73m2, and the median age-related hemoglobin standard deviation score (SDS) was -1.1 (-2.3, 0.2). Anemic subjects had higher C-terminal FGF23 SDS than non-anemic subjects (2.36 vs 1.01, p< 0.0001). In a multivariable linear regression model, log-transformed C-terminal FGF23 was independently and inversely associated with hemoglobin SDS (see Table 1). Log-transformed intact FGF23 was not significantly associated with hemoglobin SDS or serum iron. Change in hemoglobin SDS was additionally found to be inversely correlated to change in log-transformed C-terminal FGF23 over 2 years (Pearson correlation coefficient=-0.16, p=0.001). Using linear mixed effects models, longitudinal analyses over 6 years of follow up demonstrated that baseline hemoglobin SDS was inversely and independently associated with log-transformed C-terminal FGF23 (β =-0.023, p< 0.0001) (see Table 2). Likewise, baseline hemoglobin SDS had borderline significance with log-transformed intact FGF23 (β =-0.017, p=0.057).Conclusion(s): Decreased hemoglobin concentrations are independently associated with increased C-terminal FGF23 levels in pediatric CKD. This relationship is weaker with intact FGF23 levels, potentially implicating anemia-associated factors such as erythropoietin, increasing post-translational FGF23 cleavage. Further studies are warranted to identify and investigate these other influencing factors with FGF23.
Table 1: Univariable and multivariable linear regression modeling of determinants of circulating log-transformed C-terminal FGF23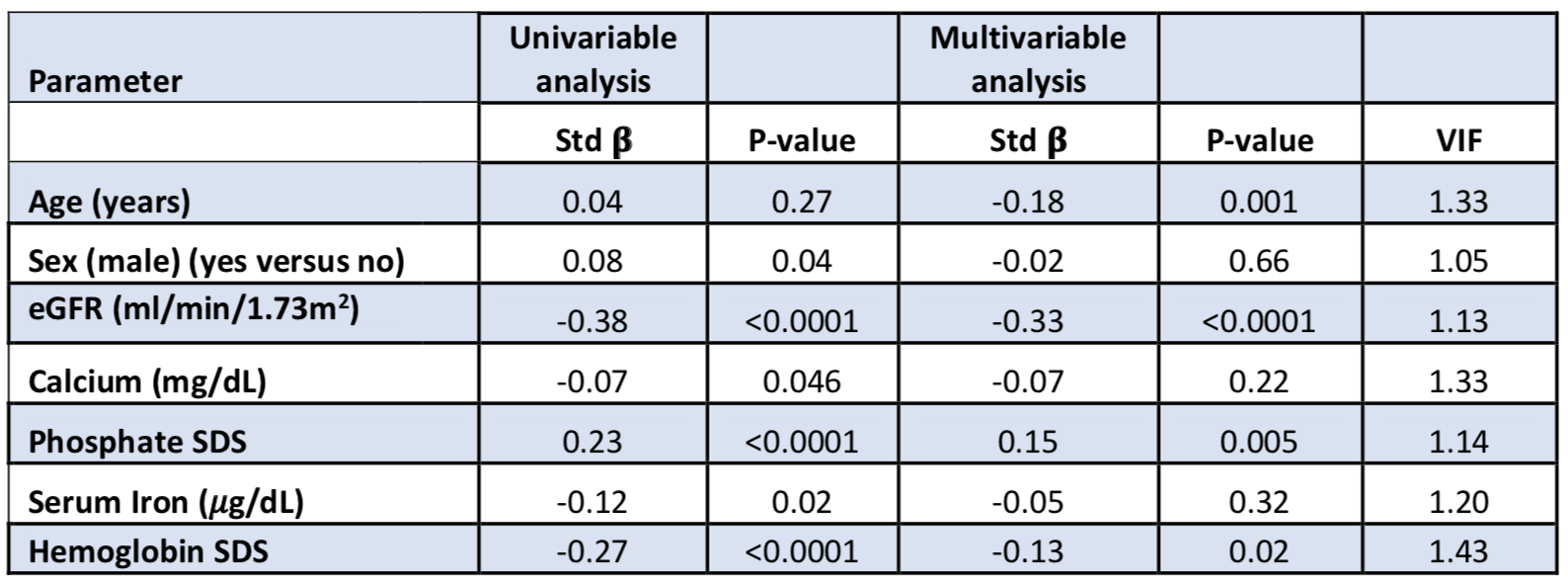
Table 2: Associations of baseline hemoglobin SDS with circulating log-transformed C-terminal FGF23 in a linear mixed effects model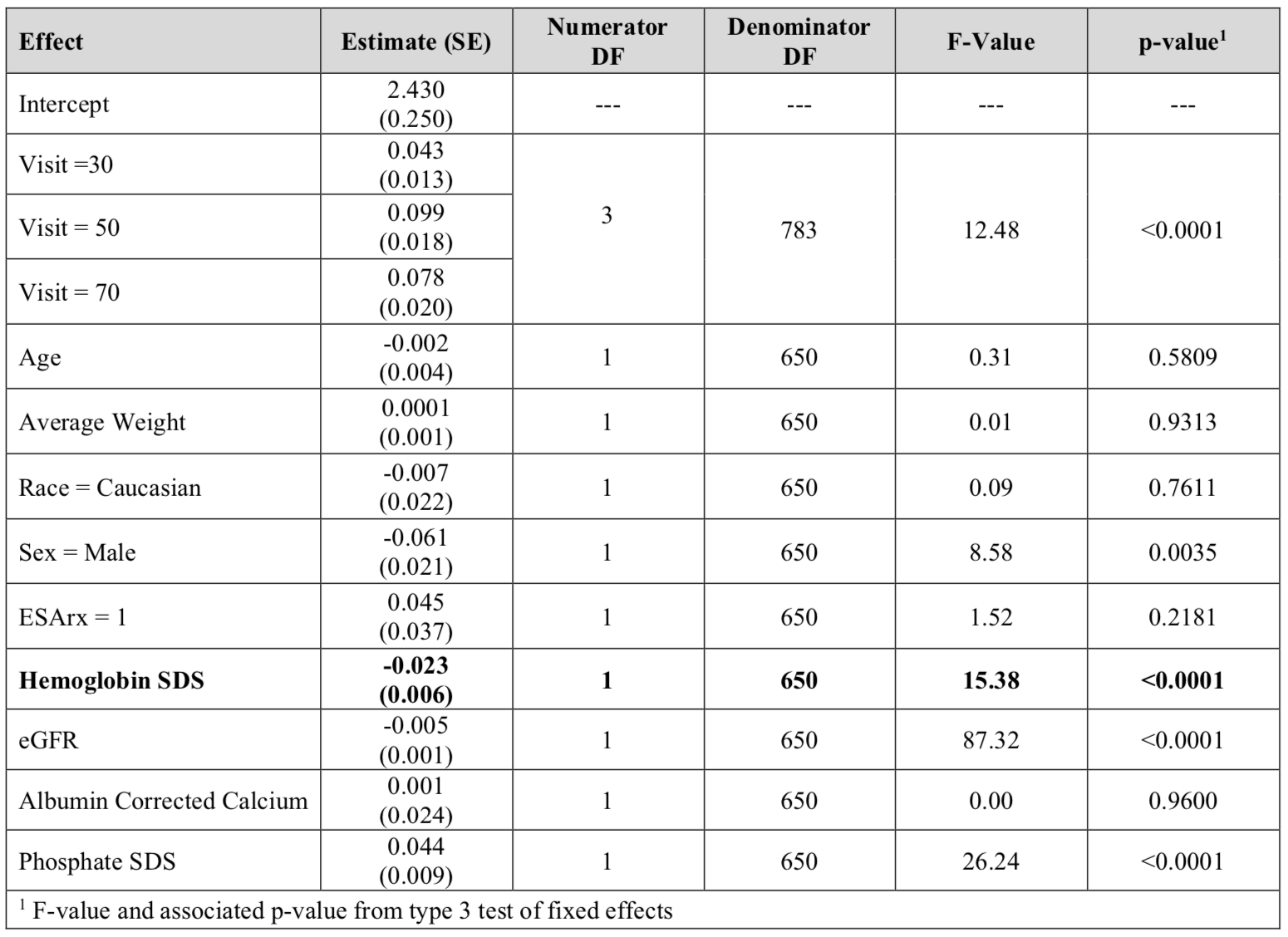
Objective: The primary aims of this study are to assess whether anemia and/or iron deficiency are associated with increased FGF23 concentrations.
Design/Methods: Anemia, iron and FGF23 profiles were characterized in cross-sectional and longitudinal analyses of subjects enrolled in the CKiD study. Participants included children aged 1 month to 16 years old with mild to moderate CKD.
Results: In a cross-sectional analysis of 686 pediatric CKD patients at baseline (median (interquartile range) age 11 (8, 15) years, 62% male, 14% Hispanic), the median estimated glomerular filtration rate (eGFR) was 55 (41, 71) ml/min/1.73m2, and the median age-related hemoglobin standard deviation score (SDS) was -1.1 (-2.3, 0.2). Anemic subjects had higher C-terminal FGF23 SDS than non-anemic subjects (2.36 vs 1.01, p< 0.0001). In a multivariable linear regression model, log-transformed C-terminal FGF23 was independently and inversely associated with hemoglobin SDS (see Table 1). Log-transformed intact FGF23 was not significantly associated with hemoglobin SDS or serum iron. Change in hemoglobin SDS was additionally found to be inversely correlated to change in log-transformed C-terminal FGF23 over 2 years (Pearson correlation coefficient=-0.16, p=0.001). Using linear mixed effects models, longitudinal analyses over 6 years of follow up demonstrated that baseline hemoglobin SDS was inversely and independently associated with log-transformed C-terminal FGF23 (β =-0.023, p< 0.0001) (see Table 2). Likewise, baseline hemoglobin SDS had borderline significance with log-transformed intact FGF23 (β =-0.017, p=0.057).Conclusion(s): Decreased hemoglobin concentrations are independently associated with increased C-terminal FGF23 levels in pediatric CKD. This relationship is weaker with intact FGF23 levels, potentially implicating anemia-associated factors such as erythropoietin, increasing post-translational FGF23 cleavage. Further studies are warranted to identify and investigate these other influencing factors with FGF23.
Table 1: Univariable and multivariable linear regression modeling of determinants of circulating log-transformed C-terminal FGF23

Table 2: Associations of baseline hemoglobin SDS with circulating log-transformed C-terminal FGF23 in a linear mixed effects model

