Back
Nephrology: Transplant
Category: Abstract Submission
Nephrology II: Transplant
42 - Safety and Efficacy of Belatacept Conversion in Adolescent and Young Adult Kidney Transplant Recipients
Saturday, April 23, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 42
Publication Number: 42.235
Publication Number: 42.235
Charu Gupta, Children's National Hospital, Silver Spring, MD, United States; christy Petyak, Children's National Health System, Washington, DC, United States; Kristen Sgambat, Children's National Health System, Silver Spring, MD, United States; Asha Moudgil, Children's National Health System, Washington, DC, United States

Charu Gupta, MBBS, MPH
Attending Physician
Children's National Hospital
Silver Spring, Maryland, United States
Presenting Author(s)
Background: Belatacept (BELA), a co-stimulation blocker, provides improved graft function and graft survival compared to calcineurin inhibitors in adult kidney transplant (KTx) recipients. Its use in adolescents and young adults is limited.
Objective: We report the safety and efficacy of BELA conversion in 9 adolescents and young adults after KTx.
Design/Methods: Nine adolescent and young adult KTx recipients were converted from tacrolimus (TAC) to BELA between 2016-2021. All patients had received induction with 4 doses of thymoglobulin and 5 doses of IV methylprednisolone. Indications for conversion included nonadherence in 3, TAC intolerance in 4, patient preference, and insulin-dependent diabetes mellitus in 1 each. BELA 5 mg/kg was given every 2 weeks for 8 weeks and monthly thereafter. TAC dose was decreased by 50 % after 2nd dose of BELA, further tapered, and discontinued after 4th dose. Other maintenance immunosuppression with mycophenolate mofetil was optimized. Data on infections (UTI and others), viral infections (CMV, EBV, BKV), hospitalizations, donor-specific antibodies (DSA), and estimated GFR was collected for 6 months prior and until the last follow up after conversion.
Results: Patient characteristics are shown in table 1. There was a decrease in infections (UTIs and others) and hospitalizations per 100 patient months of follow-up after conversion to BELA and GFR remained stable (table 2). Post conversion, 1 patient had brief CMV replication (responded to oral valganciclovir), 7 patients with preexisting low-grade EBV replication remained asymptomatic, did not show an increase in viral load, and did not develop the post-transplant lymphoproliferative disease. No patient had BK replication. There were no acute rejection (AR) episodes and no patients developed new DSA post-conversion. Three patients with preexistent DSA showed a decrease in titer. One patient with diabetes mellitus showed improved HbA1C and reduced insulin dose post-conversion.Conclusion(s): BELA conversion in adolescents and young adults is safe and effective with no evidence of increase in infections, hospitalizations, AR episodes, DSA formation or viral replication and results in stable graft function. Larger conversion studies are needed to validate these results.
Patient characteristics.jpg) yrs: Years; mo: months; Cx: Conversion to BELA; Tx: Transplant; GN: Glomerulonephritis; HUS: Hemolytic Uremic Syndrome
yrs: Years; mo: months; Cx: Conversion to BELA; Tx: Transplant; GN: Glomerulonephritis; HUS: Hemolytic Uremic Syndrome
Results data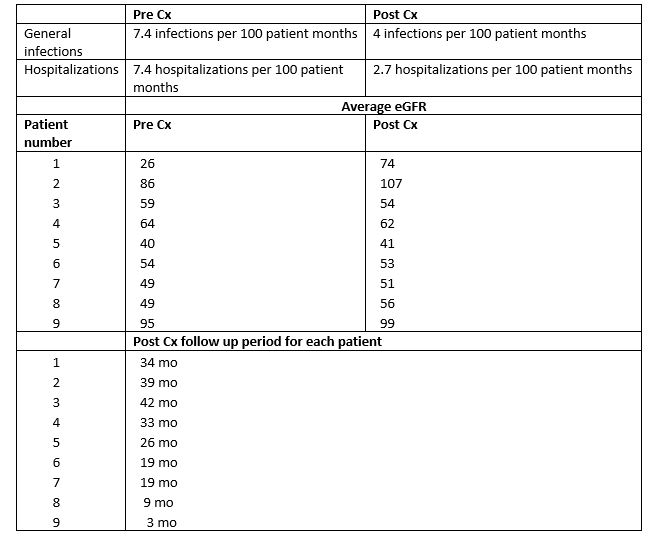 eGFR= estimated glomerular filtration rate in ml/min/1.73m2; Cx= conversion to Belatacept; mo=months
eGFR= estimated glomerular filtration rate in ml/min/1.73m2; Cx= conversion to Belatacept; mo=months
Objective: We report the safety and efficacy of BELA conversion in 9 adolescents and young adults after KTx.
Design/Methods: Nine adolescent and young adult KTx recipients were converted from tacrolimus (TAC) to BELA between 2016-2021. All patients had received induction with 4 doses of thymoglobulin and 5 doses of IV methylprednisolone. Indications for conversion included nonadherence in 3, TAC intolerance in 4, patient preference, and insulin-dependent diabetes mellitus in 1 each. BELA 5 mg/kg was given every 2 weeks for 8 weeks and monthly thereafter. TAC dose was decreased by 50 % after 2nd dose of BELA, further tapered, and discontinued after 4th dose. Other maintenance immunosuppression with mycophenolate mofetil was optimized. Data on infections (UTI and others), viral infections (CMV, EBV, BKV), hospitalizations, donor-specific antibodies (DSA), and estimated GFR was collected for 6 months prior and until the last follow up after conversion.
Results: Patient characteristics are shown in table 1. There was a decrease in infections (UTIs and others) and hospitalizations per 100 patient months of follow-up after conversion to BELA and GFR remained stable (table 2). Post conversion, 1 patient had brief CMV replication (responded to oral valganciclovir), 7 patients with preexisting low-grade EBV replication remained asymptomatic, did not show an increase in viral load, and did not develop the post-transplant lymphoproliferative disease. No patient had BK replication. There were no acute rejection (AR) episodes and no patients developed new DSA post-conversion. Three patients with preexistent DSA showed a decrease in titer. One patient with diabetes mellitus showed improved HbA1C and reduced insulin dose post-conversion.Conclusion(s): BELA conversion in adolescents and young adults is safe and effective with no evidence of increase in infections, hospitalizations, AR episodes, DSA formation or viral replication and results in stable graft function. Larger conversion studies are needed to validate these results.
Patient characteristics
.jpg) yrs: Years; mo: months; Cx: Conversion to BELA; Tx: Transplant; GN: Glomerulonephritis; HUS: Hemolytic Uremic Syndrome
yrs: Years; mo: months; Cx: Conversion to BELA; Tx: Transplant; GN: Glomerulonephritis; HUS: Hemolytic Uremic SyndromeResults data
 eGFR= estimated glomerular filtration rate in ml/min/1.73m2; Cx= conversion to Belatacept; mo=months
eGFR= estimated glomerular filtration rate in ml/min/1.73m2; Cx= conversion to Belatacept; mo=months