Back
Sedation Medicine
Category: Abstract Submission
Sedation Medicine I
560 - Clinical Characteristics of Intranasal Dexmedetomidine for Sedation in Children Undergoing Non-painful Procedures
Saturday, April 23, 2022
3:30 PM – 6:00 PM US MT
Poster Number: 560
Publication Number: 560.244
Publication Number: 560.244
Amber Rogers, Society for Pediatric Sedation, Austin, TX, United States; Nicole Baier, Santa Clara Valley Medical Center, San Jose, CA, United States; Joanna Paquin, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States; Ryan Majcina, Southern Illinois University School of Medicine, Springfield, IL, United States; Janey Phelps, University of North Carolina at Chapel Hill School of Medicine, Chapel Hill, NC, United States; April HOLLENBECK, UPMC Children's Hospital Of Pittsburgh, Allison Park, PA, United States; Carmen D. Sulton, Emory University School of Medicine, Decatur, GA, United States; Joseph Cravero, Harvard Medical School, Boston, MA, United States; Daniel S. Tsze, Columbia University Vagelos College of Physicians and Surgeons, New York, NY, United States
- DT
Daniel Tsze, MD, MPH (he/him/his)
Professor of Pediatrics in Emergency Medicine
Columbia University Vagelos College of Physicians and Surgeons
New York, New York, United States
Presenting Author(s)
Background: Dexmedetomidine (DEX) is an alpha-2 receptor agonist that can provide sedation for non-painful procedures in children and can be administered by the intranasal (IN) route. However, there is substantial variation in how IN DEX is used, and it is unclear how effective different IN DEX regimens are for different procedures.
Objective: We aimed to describe clinically-relevant outcomes (e.g. adequacy of sedation, time to clinically-meaningful endpoints, adverse events) associated with different IN DEX regimens for different types of non-painful procedures.
Design/Methods: We conducted a multicenter prospective observational cross-sectional study of children ages 2 months to 18 years receiving IN DEX sedation undergoing a non-painful procedure. The treating clinician made all medication decisions. Descriptive measures of patient and sedation characteristics, adverse events, and time endpoints were reported. The Pediatric Sedation State Scale was used to assess sedation adequacy. A multivariable regression model was used to assess associations between patient and sedation characteristics and failed sedations (i.e. received parenteral sedative and/or uninterpretable test results).
Results: We enrolled 578 patients from 7 sites. Mean age was 3 years, 217 (37.5%) were female. Procedures performed included 314 auditory brainstem response tests, 132 magnetic resonance imaging scans, 89 echocardiograms, 34 electroencephalograms, and 9 computed tomography scans. Mean total IN DEX dose was 3.3 mcg/kg (95% CI 3.2, 3.4); 230 (39.8%) received an adjunct sedative (82 received IN midazolam, 136 oral midazolam). Mean procedure duration was 45.7 min. Mean times from IN DEX administration to onset of sedation, procedure start, and discharge readiness were 33.9, 35.2, and 115 minutes, respectively. There were no patients who had a cardiovascular adverse event that required an intervention. Airway/breathing interventions administered in response to an event were jaw thrust (4 patients), repositioning (2), and blow-by oxygen (4). No patients required emergent anesthesia assistance, emergent airway placement/intubation, or admission to the hospital. There were 48 (8.3%) failed sedations. Administering IN DEX without an adjunct sedative was associated with failed sedation (OR 5.9, 95% CI 2, 17.3).Conclusion(s): IN DEX, with or without adjunct sedatives, is effective for providing adequate sedation for a large proportion of children undergoing non-painful procedures, although sedations without adjunct sedatives were associated with failed sedations. There were no serious adverse events that required an intervention.
Table 1.jpg) Intranasal dexmedetomidine doses administered for non-painful procedures in children
Intranasal dexmedetomidine doses administered for non-painful procedures in children
Figure 1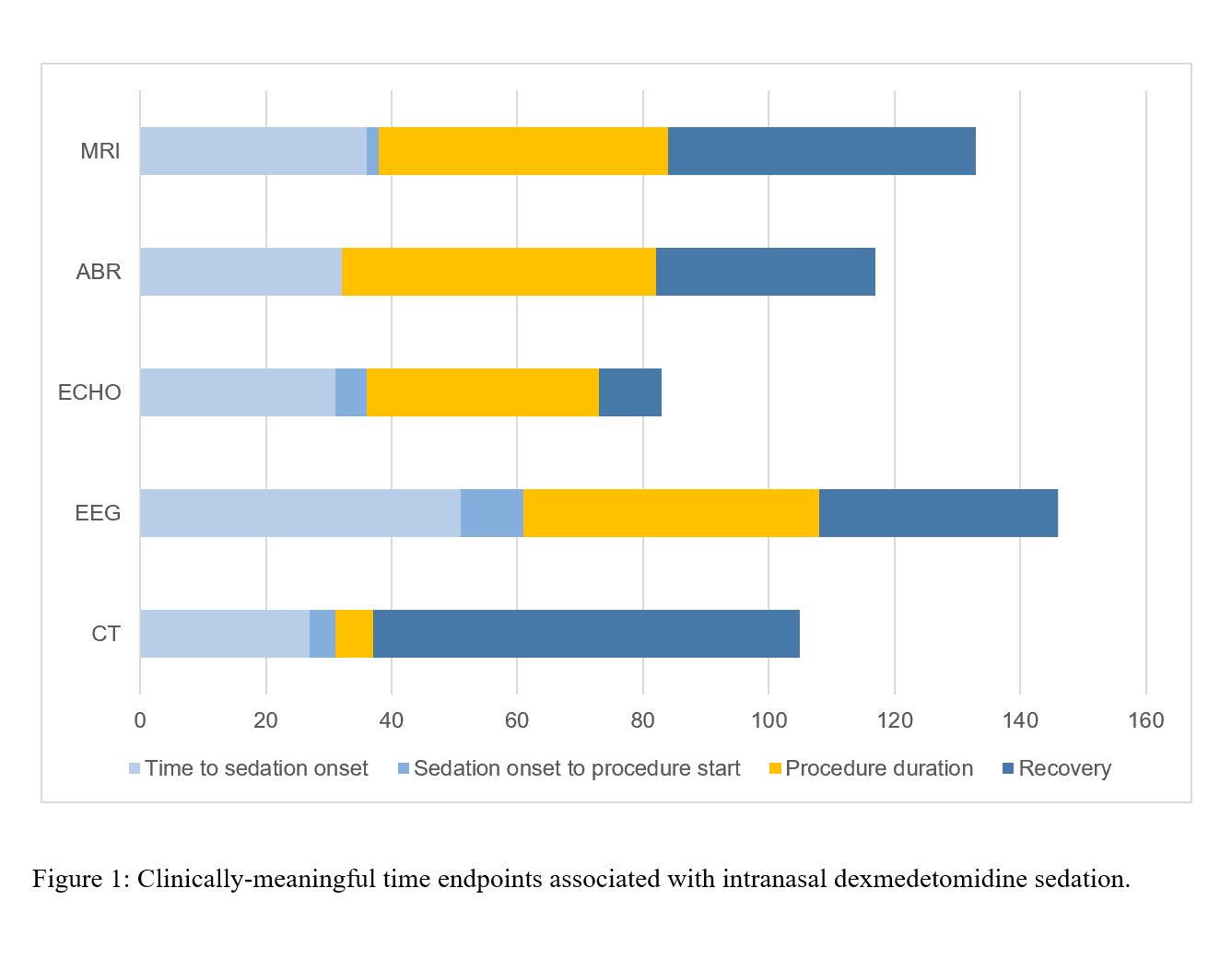 Clinically-meaningful time endpoints associated with intranasal dexmedetomidine sedation
Clinically-meaningful time endpoints associated with intranasal dexmedetomidine sedation
Objective: We aimed to describe clinically-relevant outcomes (e.g. adequacy of sedation, time to clinically-meaningful endpoints, adverse events) associated with different IN DEX regimens for different types of non-painful procedures.
Design/Methods: We conducted a multicenter prospective observational cross-sectional study of children ages 2 months to 18 years receiving IN DEX sedation undergoing a non-painful procedure. The treating clinician made all medication decisions. Descriptive measures of patient and sedation characteristics, adverse events, and time endpoints were reported. The Pediatric Sedation State Scale was used to assess sedation adequacy. A multivariable regression model was used to assess associations between patient and sedation characteristics and failed sedations (i.e. received parenteral sedative and/or uninterpretable test results).
Results: We enrolled 578 patients from 7 sites. Mean age was 3 years, 217 (37.5%) were female. Procedures performed included 314 auditory brainstem response tests, 132 magnetic resonance imaging scans, 89 echocardiograms, 34 electroencephalograms, and 9 computed tomography scans. Mean total IN DEX dose was 3.3 mcg/kg (95% CI 3.2, 3.4); 230 (39.8%) received an adjunct sedative (82 received IN midazolam, 136 oral midazolam). Mean procedure duration was 45.7 min. Mean times from IN DEX administration to onset of sedation, procedure start, and discharge readiness were 33.9, 35.2, and 115 minutes, respectively. There were no patients who had a cardiovascular adverse event that required an intervention. Airway/breathing interventions administered in response to an event were jaw thrust (4 patients), repositioning (2), and blow-by oxygen (4). No patients required emergent anesthesia assistance, emergent airway placement/intubation, or admission to the hospital. There were 48 (8.3%) failed sedations. Administering IN DEX without an adjunct sedative was associated with failed sedation (OR 5.9, 95% CI 2, 17.3).Conclusion(s): IN DEX, with or without adjunct sedatives, is effective for providing adequate sedation for a large proportion of children undergoing non-painful procedures, although sedations without adjunct sedatives were associated with failed sedations. There were no serious adverse events that required an intervention.
Table 1
.jpg) Intranasal dexmedetomidine doses administered for non-painful procedures in children
Intranasal dexmedetomidine doses administered for non-painful procedures in childrenFigure 1
 Clinically-meaningful time endpoints associated with intranasal dexmedetomidine sedation
Clinically-meaningful time endpoints associated with intranasal dexmedetomidine sedation