Nephrology: Transplant
Category: Abstract Submission
Nephrology II: Transplant
34 - COVID-19 in Pediatric Kidney Transplant: A Follow-up Report of the Improving Renal Outcomes Collaborative
Saturday, April 23, 2022
3:30 PM - 6:00 PM US MT
Poster Number: 34
Publication Number: 34.235
Publication Number: 34.235
Charles Varnell, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States; Lyndsay Harshman, University of Iowa Stead Family Children's Hospital, Iowa City, IA, United States; Michael Seifert, University of Alabama School of Medicine, Birmingham, AL, United States; David K. Hooper, Cincinnati Children's Hospital Medical Center, Cincinnati, OH, United States

Charles Varnell, MD, MS
Associate Professor of Pediatrics
Cincinnati Children's Hospital Medical Center
Cincinnati, Ohio, United States
Presenting Author(s)
Background: To date studies of children with a kidney transplant and COVID-19 are limited. The Improving Renal Outcomes Collaborative (IROC) is a learning health network in the US comprised of 36 pediatric kidney transplant programs that provides infrastructure to efficiently conduct multicenter studies.
Objective: To understand the indications for testing and short-term clinical outcomes in pediatric kidney transplant patients with COVID-19
Design/Methods: A data collection tool was created and sent to IROC centers to collect COVID-19 testing, indications, and clinical outcomes data. This data was linked to patient-specific demographic and clinical data in the IROC registry. We previously reported the results of this effort from April 6-September 3, 2020 (era 1). Herein we report data from September 4, 2020 – February 28, 2021 (era 2). Analyses were done to evaluate the differences in testing frequency and positive testing over the two study eras.
Results: In era 1, 22 centers submitted testing data; in era 2, 21 centers submitted testing data. There were 281 tests in 281 patients analyzed in era 1 and 857 tests for 513 patients in era 2. From era 1 to era 2, the proportion of positive tests increased from 24/281 (8.54%) to 135/857 (15.75%). Testing frequency and results from eras 1 and 2 are displayed in Figure 1. Figure 2 shows the frequency of positive COVID-19 tests over the same time period with the cohort split into age < 12 years and age > 12 years. 145 patients tested positive for COVID-19 over both eras, and there was no difference in the symptoms at the time of testing between the two eras. The most common symptoms for both eras were fever (35%), cough (32%), rhinorrhea (20%), vomiting/diarrhea (12%). Over both eras, 45/145 (31%) that tested positive for COVID-19 had no symptoms at the time of testing. 128 patients (88.3%) had no transplant complications. 1 (0.7%) patient had T cell-mediated rejection, 3 (2.1%) had antibody-mediated rejection, 1 (0.7%) had mixed T cell- and antibody-mediated rejection, 10 (6.9%) had acute kidney injury, and 2 (1.4%) experienced graft failure.Conclusion(s): In this cohort, there was overall more testing and more positive COVID-19 tests in era 2. The dates of era 2 correspond with children returning to school and the Winter surge in cases in the US. Despite the increased number and proportion of positive patients, the clinical outcomes are consistent with reported outcomes for their non-immunosuppressed peers. Follow-up studies will be required to evaluate whether the availability of vaccines and new variants affect clinical outcomes
Bar graph for COVID-19 testing and percent of positive tests by month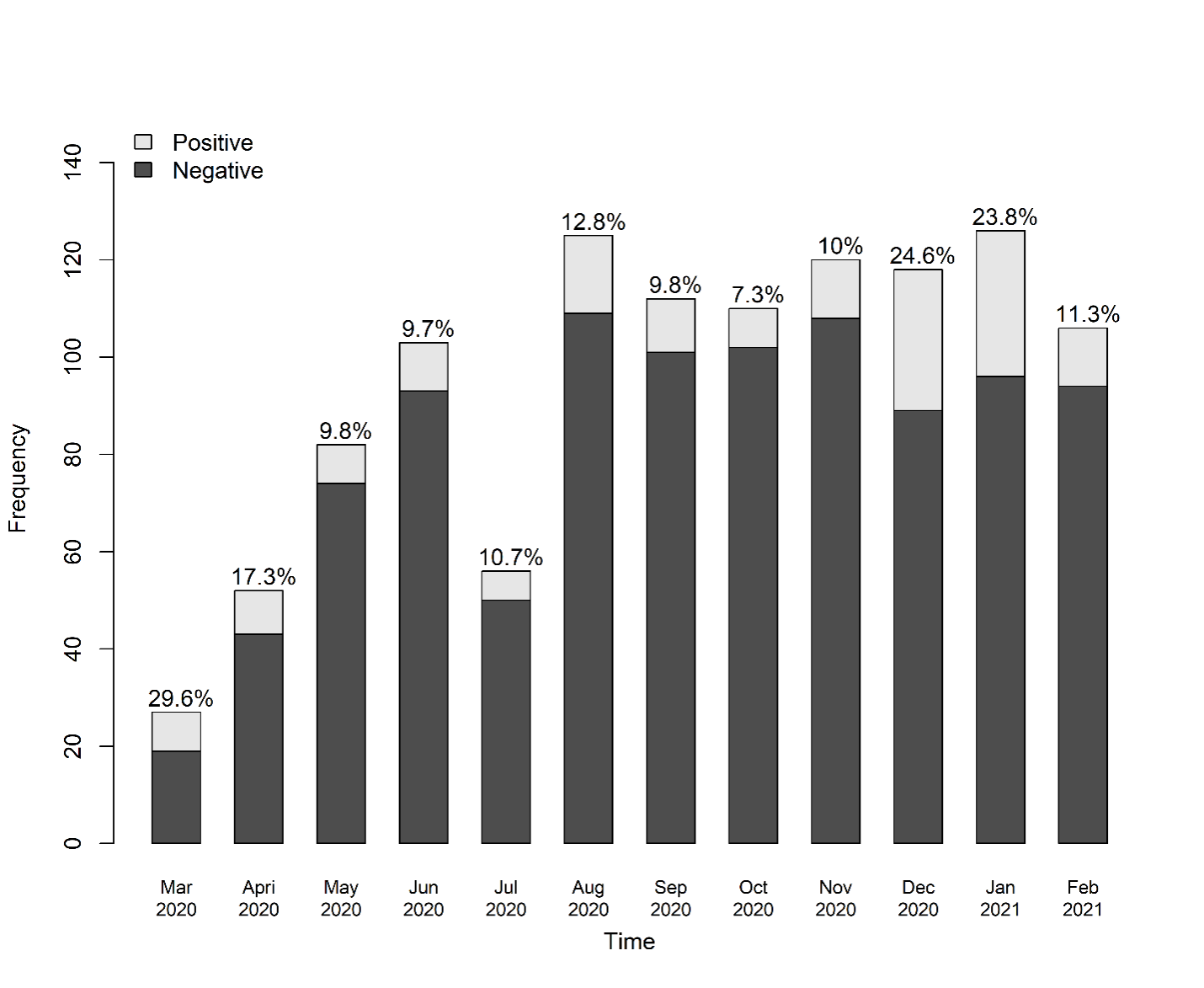
Line graph for percent positive COVID-19 tests by month by age < 12 years and age > 12 years.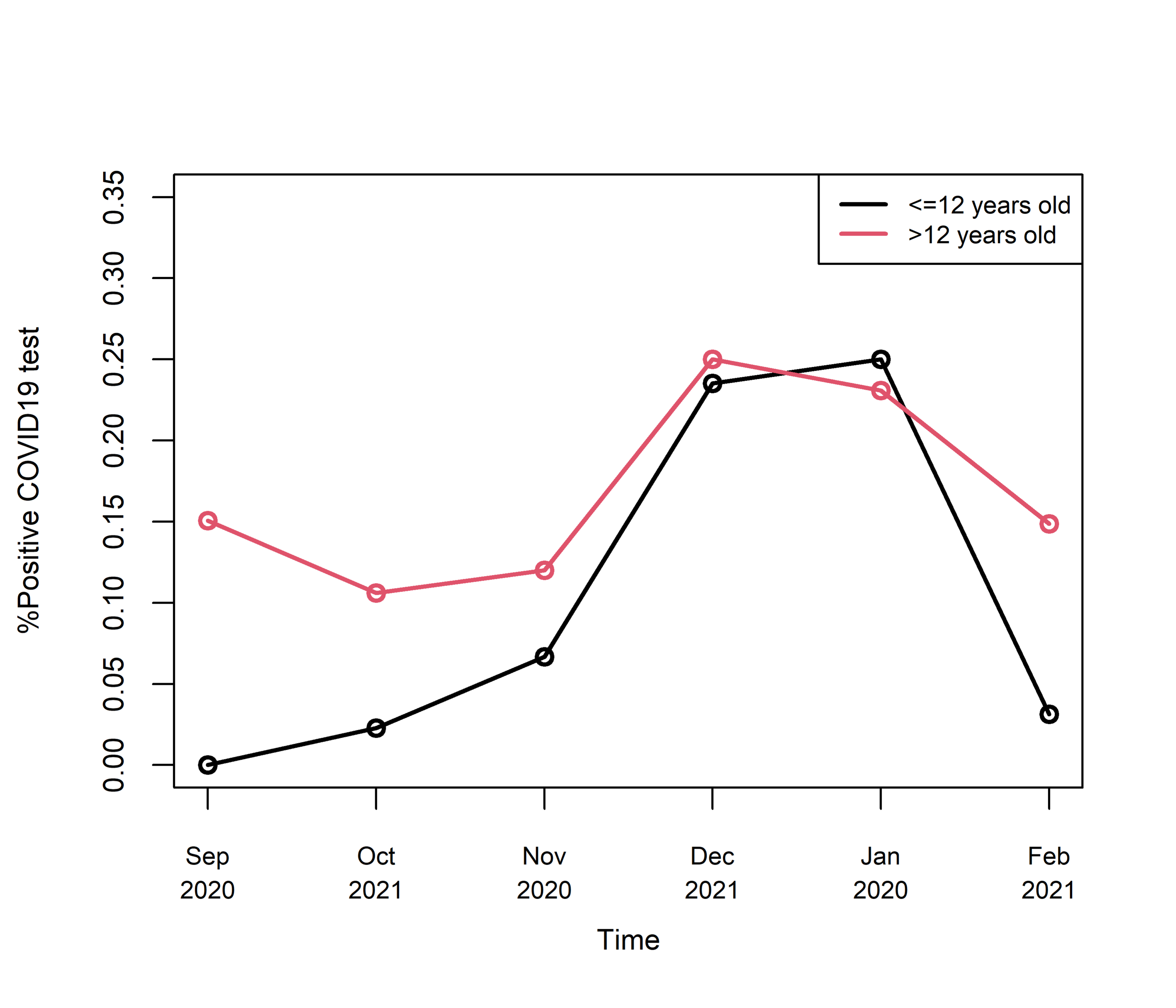
Objective: To understand the indications for testing and short-term clinical outcomes in pediatric kidney transplant patients with COVID-19
Design/Methods: A data collection tool was created and sent to IROC centers to collect COVID-19 testing, indications, and clinical outcomes data. This data was linked to patient-specific demographic and clinical data in the IROC registry. We previously reported the results of this effort from April 6-September 3, 2020 (era 1). Herein we report data from September 4, 2020 – February 28, 2021 (era 2). Analyses were done to evaluate the differences in testing frequency and positive testing over the two study eras.
Results: In era 1, 22 centers submitted testing data; in era 2, 21 centers submitted testing data. There were 281 tests in 281 patients analyzed in era 1 and 857 tests for 513 patients in era 2. From era 1 to era 2, the proportion of positive tests increased from 24/281 (8.54%) to 135/857 (15.75%). Testing frequency and results from eras 1 and 2 are displayed in Figure 1. Figure 2 shows the frequency of positive COVID-19 tests over the same time period with the cohort split into age < 12 years and age > 12 years. 145 patients tested positive for COVID-19 over both eras, and there was no difference in the symptoms at the time of testing between the two eras. The most common symptoms for both eras were fever (35%), cough (32%), rhinorrhea (20%), vomiting/diarrhea (12%). Over both eras, 45/145 (31%) that tested positive for COVID-19 had no symptoms at the time of testing. 128 patients (88.3%) had no transplant complications. 1 (0.7%) patient had T cell-mediated rejection, 3 (2.1%) had antibody-mediated rejection, 1 (0.7%) had mixed T cell- and antibody-mediated rejection, 10 (6.9%) had acute kidney injury, and 2 (1.4%) experienced graft failure.Conclusion(s): In this cohort, there was overall more testing and more positive COVID-19 tests in era 2. The dates of era 2 correspond with children returning to school and the Winter surge in cases in the US. Despite the increased number and proportion of positive patients, the clinical outcomes are consistent with reported outcomes for their non-immunosuppressed peers. Follow-up studies will be required to evaluate whether the availability of vaccines and new variants affect clinical outcomes
Bar graph for COVID-19 testing and percent of positive tests by month

Line graph for percent positive COVID-19 tests by month by age < 12 years and age > 12 years.

