Asthma
Category: Abstract Submission
Asthma
31 - A Descriptive Study of Patient Profiles and Treatment Response at Severe Asthma Clinic (SAC) at a Tertiary Care Center
Sunday, April 24, 2022
3:30 PM - 6:00 PM US MT
Poster Number: 31
Publication Number: 31.300
Publication Number: 31.300
Tatyana G. Mills, University of California, San Diego School of Medicine, San Diego, CA, United States; Suzan Mahdai, UCSD, LA MESA, CA, United States; Andrew Defante, Rady Children's Hospital San Diego, San Diego, CA, United States; Euyhyun Lee, UCSD ACTRI, San Diego, CA, United States; Jacob Parker, Rady Children's Hospital San Diego, San Diego, CA, United States; Manaswitha Khare, University of California San Diego, San Diego, CA, United States; Sydney Leibel, University of California, San Diego, San Diego, CA, United States

Tatyana Mills, MD (she/her/hers)
Resident
Rady Children's Hospital San Diego
San Diego, California, United States
Presenting Author(s)
Background: Asthma is the leading cause of chronic disease in the pediatric population. Rady Children's Hospital in San Diego has a prevalence of asthma of 8.6% compared to 8.1% nationally. Severe Asthma Clinic (SAC), a multidisciplinary clinic for patients with 2 or more ED visits in the past 6 months or 2 or more hospitalizations in the previous year, was started in 2015 with a goal to reduce asthma healthcare utilization and improve quality of life for patients with severe asthma.
Objective: The objective of this study is to 1) describe the SAC patient demographics 2) assess frequency of emergency department (ED) visits, hospitalizations, and systemic steroid prescriptions after SAC visit 3) investigate changes in Asthma Therapy Assessment Questionnaire (ATAQ) scores and pulmonary function tests (PFTs) with each SAC visit.
Design/Methods: Retrospective analysis was performed on patients seen at the multidisciplinary SAC at RCHSD from January 2015 to December 2020 three years prior to (pre-SAC) and one year after (post-SAC) the patient’s first SAC visit. Measures were compared using paired Wilcoxon Signed-Rank tests and a linear mixed effects model with random intercept structure was used to analyze change over time (ATAQ scores and PFT results). Results are reported in median and quartile (Q1-Q3) format.
Results: A total of 153 patients were included with a median age of 9.85 (6.57- 13.80) at the first SAC visit. Twenty-one percent (21.7%) were obese, 50% (76 patients) were white, 67.3% had Medicaid/Medical insurance coverage, and 43.8% had a diagnosis of allergic rhinitis (Table 1). The median household income was $64,940 based on zip code. There was a statistically significant difference (p < 0.001) for all treatment and hospital related outcomes from pre-SAC and post-SAC, including decrease in ED visits, hospitalizations, ICU admissions, number of systemic steroid courses, and inhaled corticosteroid potency scores (Table 2). With increase in time by 1-month post-SAC there was improved asthma control (decrease in ATAQ score by -0.050) and improved lung function (increase in FEV1 by 0.015 and increase in FVC by 0.020) (Figure 1). Data for post-SAC variables overlapped with the COVID-19 pandemic (after 3/15/2020) for 48 patients. COVID-19 pandemic did not significantly affect study outcomes.Conclusion(s): Multidisciplinary care at the SAC has led to a significant decrease in healthcare utilization (ED visits, hospitalizations) and improved asthma control for patients with severe asthma.
Patient Demographics.jpg) Total of 153 patients were included. Only 152 patients reported race, zip code (household income) and BMI data. All data was collected from the first severe asthma clinic (SAC) visit. Data was represented with median (Q1-Q3) format or count (%). Estimated household income (HHI) was calculated based on zip code. BMI percentile was categorized using the CDC percentile categories. Past medical history was identified based on patient problem list ICD 9/10 codes at the first SAC visit. Smoking history was collected by screening assessments in the inpatient or ambulatory setting 1 year prior to the first SAC visit.
Total of 153 patients were included. Only 152 patients reported race, zip code (household income) and BMI data. All data was collected from the first severe asthma clinic (SAC) visit. Data was represented with median (Q1-Q3) format or count (%). Estimated household income (HHI) was calculated based on zip code. BMI percentile was categorized using the CDC percentile categories. Past medical history was identified based on patient problem list ICD 9/10 codes at the first SAC visit. Smoking history was collected by screening assessments in the inpatient or ambulatory setting 1 year prior to the first SAC visit.
Change in Treatment and Hospital Related Outcomes from Pre-SAC to Post-SAC Visit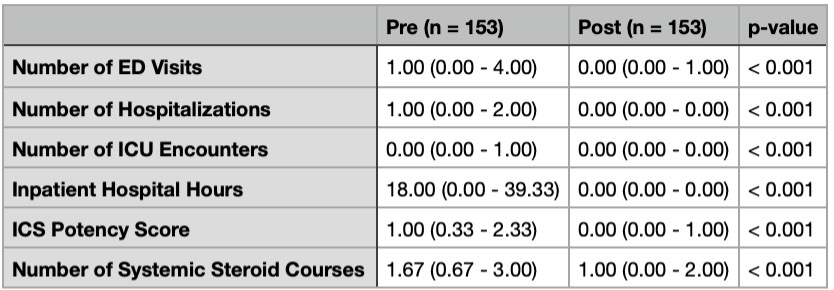 ED: Emergency Department; ICU: Intensive Care Unit; ICS: inhaled corticosteroid.
ED: Emergency Department; ICU: Intensive Care Unit; ICS: inhaled corticosteroid.
Table 2 shows data analysis using the Paired Wilcoxon Signed-Rank Test to compare a mean of the 3 years prior and 1 year after first SAC visit. Data was reported in a median (Q1-Q3) format. ED visits included patients that were discharged from the ED and those later admitted to the hospital. Inpatient hospital hours included entire period of inpatient admission including the intensive care unit (ICU). Each ICS prescription was given a potency score of mild, medium, or high (1, 2, 3) based on the 2007 NIH Asthma guidelines. Combination ICS/long acting beta-agonist were also given a potency score mild, medium, or high (1,2,3) based on the 2020 AAP Asthma Guidelines Update. Systemic steroid courses included the number of either inpatient or outpatient prescribed steroid courses, days varied, for an asthma exacerbation.
Objective: The objective of this study is to 1) describe the SAC patient demographics 2) assess frequency of emergency department (ED) visits, hospitalizations, and systemic steroid prescriptions after SAC visit 3) investigate changes in Asthma Therapy Assessment Questionnaire (ATAQ) scores and pulmonary function tests (PFTs) with each SAC visit.
Design/Methods: Retrospective analysis was performed on patients seen at the multidisciplinary SAC at RCHSD from January 2015 to December 2020 three years prior to (pre-SAC) and one year after (post-SAC) the patient’s first SAC visit. Measures were compared using paired Wilcoxon Signed-Rank tests and a linear mixed effects model with random intercept structure was used to analyze change over time (ATAQ scores and PFT results). Results are reported in median and quartile (Q1-Q3) format.
Results: A total of 153 patients were included with a median age of 9.85 (6.57- 13.80) at the first SAC visit. Twenty-one percent (21.7%) were obese, 50% (76 patients) were white, 67.3% had Medicaid/Medical insurance coverage, and 43.8% had a diagnosis of allergic rhinitis (Table 1). The median household income was $64,940 based on zip code. There was a statistically significant difference (p < 0.001) for all treatment and hospital related outcomes from pre-SAC and post-SAC, including decrease in ED visits, hospitalizations, ICU admissions, number of systemic steroid courses, and inhaled corticosteroid potency scores (Table 2). With increase in time by 1-month post-SAC there was improved asthma control (decrease in ATAQ score by -0.050) and improved lung function (increase in FEV1 by 0.015 and increase in FVC by 0.020) (Figure 1). Data for post-SAC variables overlapped with the COVID-19 pandemic (after 3/15/2020) for 48 patients. COVID-19 pandemic did not significantly affect study outcomes.Conclusion(s): Multidisciplinary care at the SAC has led to a significant decrease in healthcare utilization (ED visits, hospitalizations) and improved asthma control for patients with severe asthma.
Patient Demographics
.jpg) Total of 153 patients were included. Only 152 patients reported race, zip code (household income) and BMI data. All data was collected from the first severe asthma clinic (SAC) visit. Data was represented with median (Q1-Q3) format or count (%). Estimated household income (HHI) was calculated based on zip code. BMI percentile was categorized using the CDC percentile categories. Past medical history was identified based on patient problem list ICD 9/10 codes at the first SAC visit. Smoking history was collected by screening assessments in the inpatient or ambulatory setting 1 year prior to the first SAC visit.
Total of 153 patients were included. Only 152 patients reported race, zip code (household income) and BMI data. All data was collected from the first severe asthma clinic (SAC) visit. Data was represented with median (Q1-Q3) format or count (%). Estimated household income (HHI) was calculated based on zip code. BMI percentile was categorized using the CDC percentile categories. Past medical history was identified based on patient problem list ICD 9/10 codes at the first SAC visit. Smoking history was collected by screening assessments in the inpatient or ambulatory setting 1 year prior to the first SAC visit.Change in Treatment and Hospital Related Outcomes from Pre-SAC to Post-SAC Visit
 ED: Emergency Department; ICU: Intensive Care Unit; ICS: inhaled corticosteroid.
ED: Emergency Department; ICU: Intensive Care Unit; ICS: inhaled corticosteroid. Table 2 shows data analysis using the Paired Wilcoxon Signed-Rank Test to compare a mean of the 3 years prior and 1 year after first SAC visit. Data was reported in a median (Q1-Q3) format. ED visits included patients that were discharged from the ED and those later admitted to the hospital. Inpatient hospital hours included entire period of inpatient admission including the intensive care unit (ICU). Each ICS prescription was given a potency score of mild, medium, or high (1, 2, 3) based on the 2007 NIH Asthma guidelines. Combination ICS/long acting beta-agonist were also given a potency score mild, medium, or high (1,2,3) based on the 2020 AAP Asthma Guidelines Update. Systemic steroid courses included the number of either inpatient or outpatient prescribed steroid courses, days varied, for an asthma exacerbation.
